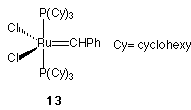| [Molecules: 5] [Related articles/posters: 062 114 036 004 067 ] |
In this poster we report for the first time our recent results from an extension of the above work to the synthesis and ROMP of monomers derived from biologically active peptides (penicillin and cephalosporin), as well as to monomers containing nucleic acid bases.
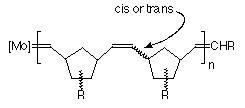
Figure 1 General structure of the polymer formed by metathesis of a norbornene
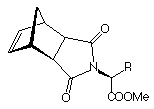
Figure 2 Structure of the amino acid derived monomers
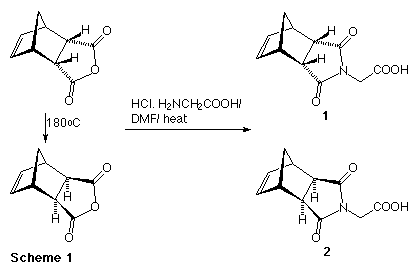
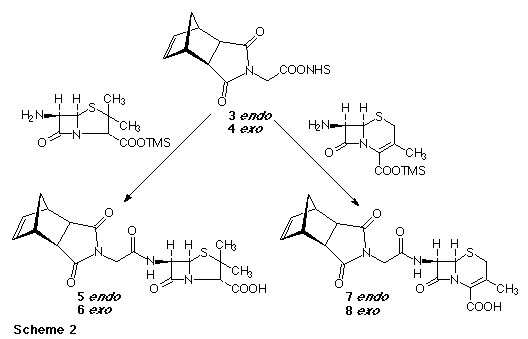
The synthesis of the penicillin and cephalosporin derived monomers started from the endo (1) and exo (2) glycine derivatives prepared as previously reported4 (Scheme 1). Compounds (1) and (2) could be converted into the corresponding acid chlorides (oxalyl chloride/ DMF) or N-hydroxysuccinimide esters (3,4) (DCC/ N-hydroxysuccinimide), with the latter giving superior results in subsequent reactions. All attempts to couple the activated glycine derivatives directly to 6-amino-penicillinic acid (6-APA), or to 7-amino-cephalosporic acid (7-ACA) were unsuccessful. However, pre-treatment of the 6-APA or 7-ACA with bis(trimethylsilylacetamide) to protect the acid functionalities as the trimethylsilyl esters followed by addition of compound (3) or (4) did give the desired monomers (5-8) after an aqueous workup as shown in Scheme 2.
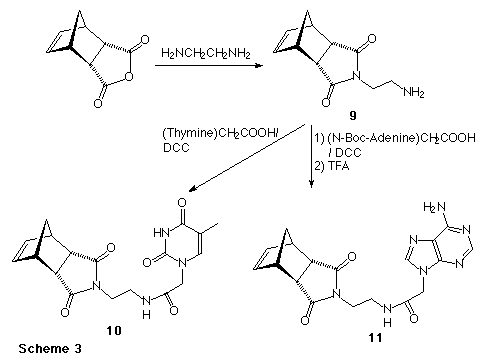
The synthesis of the endo isomers of the nucleic acid base (thymine or adenine) derived monomers proceeded as shown in Scheme 3. Thus condensation of the endo-anhydride with an excess of ethylene diamine gave amino derivative (9). Condensation of (9) with either thymine acetic acid or N-Boc-adenine acetic acid, (followed by removal of the Boc group from the adenine with trifluoroacetic acid) gave the desired norbornene derivatives (10) and (11).
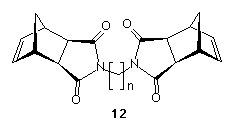
Interestingly, attempts to extend this chemistry to the exo-isomers of compounds (10) and (11) failed at the first step, as dimers of structure (12) were the only isolated products no matter how large an excess of diamine was used, and independent of the chain length of the diamine. Work is currently in progress to prepare the exo-norbornene derivatives utilising partially protected diamines.
Studies on the ROMP of monomers (5-8) and (10-11) are currently underway, using both molybdenum and ruthenium based initiators. Compound (6) has been successfully polymerised, and the resulting polymer characterised by electrospray mass spectrometry. When 10 equiv. of monomer (6) were added to a solution of the ruthenium based initiator (13), electrospray mass spectrometry showed the presence of molecular ion peaks corresponding to oligomers containing 8-15 monomer units. Similarly, thymine derived monomer (10) could also be polymerised using (13) as the initiator and electrospray mass spectrometry showed the presence of species containing between four and ten monomer units. Further work on these polymerisation reactions is currently in progress.