| [Related articles/posters: 051 071 023 ] |
Carbon centered radicals obtained, by action of stannylhydrides,
from sp2 carbon halides, have been widely used to establish
a new C - C bond 1.
Although, usually Calkyl-Calkyl
or Caryl-Calkyl
bonds are formed by this method, it has also been occasionally used also
in the formation of Caryl-Caryl
bonds 2.
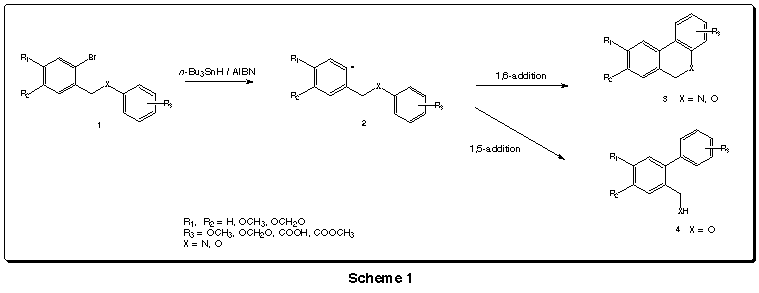
We have systematically studied additions of such aryl radicals derived
from N-o-bromobenzylanilines 3
and o-bromobenzyl phenyl ethers 4
to furnish biaryl compounds.
The first compounds exclusively suffered 1,6-addition,
leading to the corresponding phenanthridines. The latter gave the two possible
1,6-cycloadditions, leading to dibenzopyranes, and 1,5-cycloaddition, affording
phenyl benzyl alcohols. The extention of each process deppends on the aromatic
substitution R3 (Scheme 1).
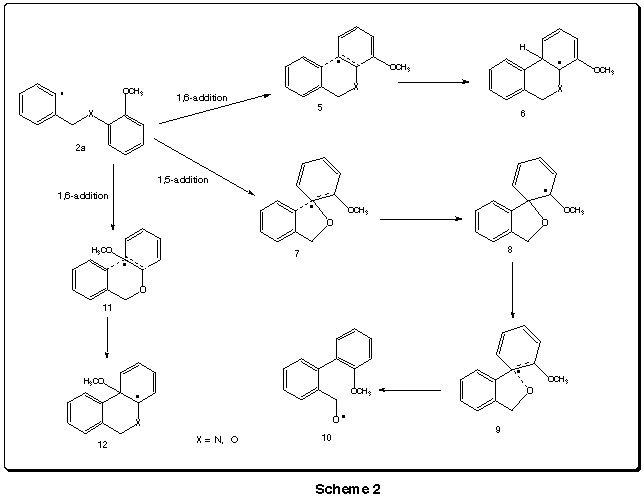
In order to understand such different behaviour
in so similar systems, molecular moddeling was performed using MOPAC (version
3.7) at the PM3 level. As it would be interesting to predict how analogue
sulfur compounds would behave under the same conditions, calculations were
run for them as well. Heats of formation were determined for species 2,
6, 8, 10 and 12
and the corresponding transition states 5, 7
,9 and 11 (Scheme 2). The results are
presented on Table 1 and ploted
on Graphics 1 - 3.The
geometries of the above species (X = N) are shown in the animation.
2. Results
3. Discussion
From Graphic 1, it seems that for nitrogen radical 2, the energy barrier leading to the 1,6-additiond product 6 is much lower than the energy barriers for compounds 8 and 12, being these thermodinamically less stable than compound 6. It is also apparent that, even if 8 is formed, the energy barrier for its conversion to 10 is too high.
Graphic 2 shows that for oxygen radical 2, some competition between the three processes is to be expected. As a matter of fact, the energy barriers leading to compounds 6 and 8 have approximate values and, although the barrier for compound 8 formation is somewhat higher than for 6, its thermodinamical stability is comparable to the one of compound 12. In this case, obtention of the transition state 9, leading to 10, is expected to be easier than for the nitrogen analogue.
Graphic 3 suggests that for the sulfur radical 2, one would expect a larger competition between 1,5-addition, leading to 8, and 1,6-additin leading to 6, than in the previous cases studied. Formation of 8 can easily lead to 10. It is possible that formation of 12 occurs, but to a less extent than in the oxygen analogue. The barrier leading to it, is the highest of all and this species is the thermodinamically least stable.
The experimental results for X=N and X=O are in accordance
with these calculations.
1 - B. Giese, Radicals in Organic Synthesis;
Formation of C-C Bonds, Pergamon Press, New York, 1986
2 - a) N. S. Narasimhan, I. S. Aiden, Tetrahedron
Lett., 1988, 29, 2987; b) H. Togo, O. Kikuchi, Tetrahedron
Lett., 1988, 29, 4133; c) W. B. Motherwell, A. M. K.
Pennell, J. Chem. Soc., Chem. Commun., 1991, 877
3 - A. M. Rosa, A. M. Lobo, P. S. Branco, S. Prabhakar,
A. M. D. L. Pereira, Tetrahedron , 1997, 53, 269
4 - A. M. Rosa, A. M. Lobo, P. S. Branco, S. Prabhakar,
Tetrahedron, 1997, 53, 285
Acknowledgements
We thank Fundação para a Ciência e
Tecnologia (FCT, Lisbon), PRAXIS and FEDER programs for partial finantial
support. A. M. R. thanks PRAXIS XXI for a post-doctoral grant and Doctor
Carlota Conesa-Moratilla for her valuable help.