Email Discussion: Re: 65:message to Ganesh Pandey sankar
This paper reports the construction of spirocyclic systems by direct Intramolecular nucleophilic substitution by silylenol ethers to the PET (Photoinduced Electron Transfer) generated arene radical (e.g., 1 to 2). The success in the transformation of 1 to 2 encouraged us to attempt the synthesis of spirocyclic part of Fredericamycin A.

Carboannulation processes are among the most important reactions in organic synthesis[1]. Many natural products have been characterized as possing spiro linkages. Various synthetic methods were available for the construction of spirocyclic center. Common reactions such as carbene or carbenoid additions to exocyclic olefins, cycloadditions, acyloin condensations, acylation, Dieckmann cyclization etc., [2] which have found application for the preparation of spirocompounds. The recent reports on the spirocyclization reactions using Pd(0), Lewis acid etc., were well documented[3] We have previously reported that ketone generated silyl enol ethers can be used as efficient carbon nucleophiles, in the intramolecular nucleophilic aromatic substitution reactions, involving PET generated arene radical cation intermediate [4].
Scheme- 1 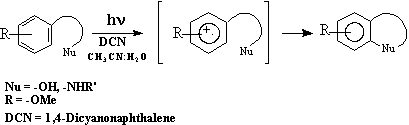
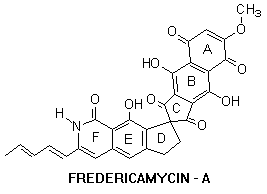
The present study upsurge our preliminary success and generalize this strategy in the synthesis of spirocyclic compounds. These spirocyclic compounds are structurally related to antitumor antibiotic Fredericamycin A , isolated from Streptomyces griseus, bearing a unique spiro[4.4]nonene center.
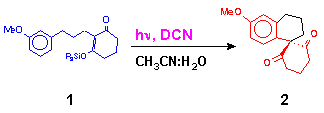
Our approach on the construction of spirocyclic skeleton involves preparation of suitable silyl enol ethers of the type 1 as reported.[5] followed by photolysis reaction using DCN (1,4 - Dicyanonaphthalene) as the electron acceptor in acetonitrile. The examples with ketone generated silyl enol ethers (entry 1-3) were included to probe the substituent effects on the reactivity and regioselectivity.
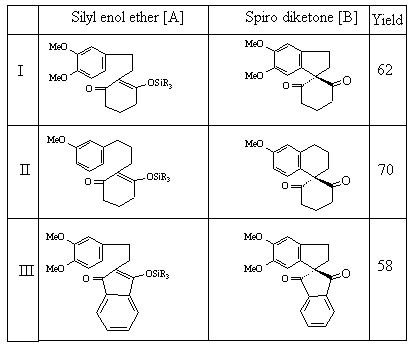
The regiospecificity of these cyclizations are in accordance with the calculated electron densities (Huckel or MNDO) at different carbons of the HOMO of the arene radical cations as observed earlier [6]. The attractive feature of this strategy lies in its ability to alter the size of newly formed spiro[n.m]cyclic ring in the product. The following figure shows the geometry of the molecule varied to obtain the minimum possible potential energy.
A typical photochemical reaction procedure consists of the irradiation of a mixture of silyl enol ethers (2 mmol) and DCN 0.06 g (0.34 mmol) in 500 ml of CH3CN:H2O (4:1) for 3-4 h through pyrex filtered light (>280 nm all light absorbed by enol ether only) using 450 W Hanovia lamp without removing the dissolved oxygen. Excess irradiation time should be avoided to control side reactions leading to secondary products. The progress of the reaction is monitored by TLC and GC. The irradiation is discontinued immediately after the complete consumption of starting silyl enol ether. Removal of the solvent and chromatographic purification of the reaction mixture gave spirocyclic products I-III [B] and characterised by 1H NMR, 13C NMR and Mass spectral data. DCN was recovered quantitatively (98 %) at the end of the reaction [7]. During the irradiation of silyl enol ethers ,minor quantity (~10 %) of starting precursor ketones are also formed which has been shown to be formed by the thermal reversion of silyl enol ethers by adequate control experiment.
Acknowledgement:
M.K thankful to CSIR,New Delhi for the award of Senior Research Fellowship, and also to Director, NCL for providing infrastructure facilities.