Email Discussion:
Poster 35 (Dr. Orera)
Pedro Merino
Email Discussion:
Re: Poster 35 (Prof. Orena)
orena@anvax1.cineca.it
Email Discussion:
http://www.ch.ic.ac.uk/papers/orena
Yannick LANDAIS
Email Discussion:
re: Landais query for poster 35 (Orena)
orena@anvax1.cineca.it


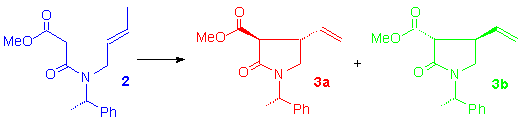
In order to obtain either 1a or 1b, the amide 2, containing the (S)-phenylethylamine as the chiral auxiliary, has been cyclised with Mn(OAc)3-Cu(OAc)2. A diastereomeric mixture (d.r. 70:30) of pyrrolidin-2-ones 3a and 3b is obtained in 53% yield. After silica gel chromatography, pure 3a and 3b are isolated and the configurations are assigned on the basis of 1H NMR spectra and confirmed by n.O.e. experiments. The moderate distereoselection can be explained through molecular mechanics calculations.
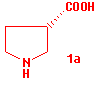
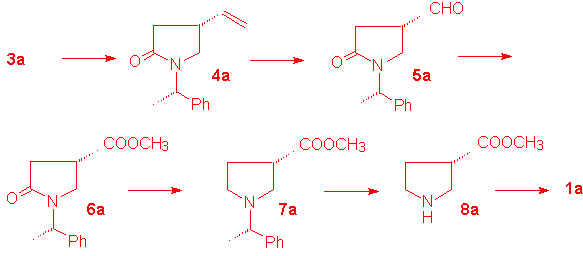
The major isomer 3a is readily converted into the 4-ethenylpyrrolidin-2-one (4a) in 75% yield by treatment with NaCl in wet DMF for 2 h. This compound undergoes ozonization in dichloromethane at -78 °C to give the aldehyde 5a in 78% yield. No trace of epimerization at C-4 is evidenced by 1H NMR spectrum. By oxidation with Jones' reagent at 0 °C the aldehyde 5a is converted into the corresponding carboxylic acid which, after treatment with diazomethane in methanol, affords the methyl ester 6a in 68% yield. The amidic carbonyl is removed by reaction with borane-dimethylsulphide in THF at 45 °C and the aminoester 7a is recovered in 75% yield after silica gel chromatography. The benzylic group at nitrogen is removed by catalytic hydrogenation on Pd-C 10% in methanol. The solvent containing the ester 8a is evaporated and the residue is placed into a column fitted with Amberlyst IRA 400 in the OH- form. After elution with acetic acid pure 1a is obtained in 65% yield as a white solid. m.p. 187-188 °C (lit. 188-190 °C); [alpha]D +18.0 (c 1, H2O) [lit. + 18.8 (c 3.1, H2O)]
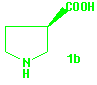
It is worth mentioning that, starting from the pyrrolidin-2-one 3b, (R)-3-pyrrolidinecarboxylic acid 1b can be obtained in the same way and comparable yield.