Curly arrow pushing is one of the essential tools of a mechanistic chemist. Many a published article will speculate about the arrow pushing in a mechanism, although it is becoming increasingly common for these speculations to be backed up by quantitative quantum mechanical and dynamical calculations. These have the potential of exposing the underlying choreography of the electronic dance (the order in which the steps take place). The basic grammar of describing that choreography tends to be the full-headed curly arrow for closed shell systems and its half-barbed equivalent for open shell systems. An effectively unstated and hence implicit rule for closed shell systems is that only one curly arrow is used per breaking or forming bond, i.e. electrons move around bonds in pairs. So consider the following reaction (inspired by a posting on Steve Bachrach’s blog)
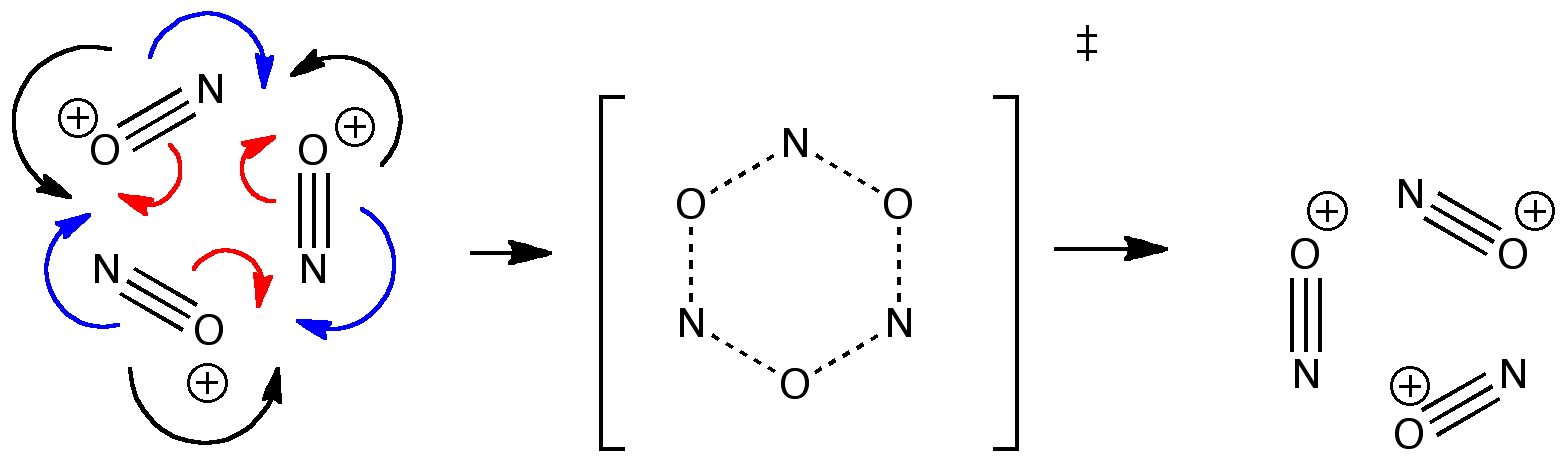
This is very much a hypothetical mechanism, or a thought-experiment if you will. Three nitrosonium cations decide to get together to swap their partners. Each diatomic molecule swaps e.g. one oxygen for another during this exchange reaction (it could easily be studied experimentally of course using isotopic substitution). Three sets of three curly arrows have been used, shown in different colours above. One set of these arrows at least has plenty of analogy in the real world; representing a π2s+π2s+π2s cycloaddition reaction. The other two sets represents rotation of the in-plane π-set and the in-plane σ-set. What about the choreography? Can all three sets move at the same time? If so, they would provide an exception to the rule above; three bonds would concurrently change their order from 3 to 0; the other three the reverse of 0 to 3.
What does quantum mechanics say about this? Well, a well defined, synchronous concerted transition state can indeed be found (B3LYP/6-31G(d), DOI: 10042/to-2905) It has one imaginary frequency (click on the above diagram to view the animation) which does indeed perform the bond transposition function required! It has the form of the so-called Kekule mode (deriving from a mode found in benzene which involves shortening of the lengths of three bonds, and lengthening of the other three, much in the manner of the resonance named after Kekule; see e.g. DOI: 10.1039/B911817A for more details). Of course, describing it as a change in the bond orders 3 → 0/0 → 3 is simplistic; the bond order in the nitrosonium cation itself is almost certainly somewhat less than three. But clearly, the implicit rule that mechanistic arrow pushing should not involve more than one arrow departing from or arriving at any one bond can be broken. I will leave it to the reader of this blog to see what happens when you try to rearrange the choreography of the above reaction. Try pushing first one set of three arrows, then another and a final third. What do you get? (the why of the dance is almost certainly due to electrostatic repulsions between the three nitrosonium cations).
Tags: animation, closed shell systems, Interesting chemistry, mechanistic chemist, open shell systems, pericyclic, Steve Bachrach
[…] previous post posed the question; during the transformation of one molecule to another, how many electron pairs […]
[…] π-set). Notice how this itself contrasts with a similarly aromatic system, the atom swapping in three nitrosonium cations, where the Kekulé mode force constant was strongly […]
[…] hard rule, but in fact its not difficult to come up with reactions which break it. For example, this one, where as many as three arrows either start or end at a bond. And, as a challenge, can you break […]
[…] But could it be four, as shown in the above example? Or perchance even six as discussed in this post? OK, the question is rather loaded for this example, since being a pericyclic reaction it has a […]
Nice post which the other two sets represents rotation of the in-plane π-set and the in-plane σ-set. In which the implicit rule that mechanistic arrow pushing should not involve more than one arrow departing from or arriving at any one bond can be broken. Thanks a lot for posting this article.
[…] uneventful. I conclude that if I ever see a student exam script showing two curly arrows heading directly towards the same bond at the same time, it might even deserve to be graded […]
Dear Henry,
What a thought-provoking example!
Regarding the choreography: in somewhat similar (but intramolecular) reactions of alkynes (e.g., the Bergman cyclization), the initial bonding changes usually involve the in-plane pi-system. The out-of-plane system remains a bystander for quite a long time until the relatively short incipient C…C distance is reached (in the TS vicinity).
However, the present example is an intermolecular process. Because it involves electronic clouds of cylindrical symmetry, one can imagine that both pi-clouds may react at the same time via banana-type overlap and two new bonds would form and break simultaneously at each of the reacting atoms.
A harder question is how the sigma C-C bonds get involved. Could it be that it is the nitrogen lone pair that interacts with the sigma*(O-N) in an SN2-like fashion to account for the last bond formation? If this is so, then the arrow pushing should change and include the lone pairs.
Also, what would you make of the fact the transition state is not planar?
Igor,
The TS is indeed not planar, but only slightly so.
A fascinating question is whether one should invoke lone pairs as “catalysts”, i.e. they are present at start and end, but perhaps actively participate at the TS. We have one such example about to appear in Chem. Comm; when it does, I will flag it here.
I tend to the answer that yes, “catalytic” lone pairs probably are involved, and yes, if we have evidence for this, we should include them in our arrow pushing.
[…] One can contrive an even higher-order reaction (thus far un-named) in which (formally) three bonds break and three bonds form at a single […]
Shown below is the IRC for this reaction. Note the change in (formal) bond order from three to zero for any given N-O pair.
The example is also probably is a good candidate to claim a record for the height of the single-step reaction barrier!
