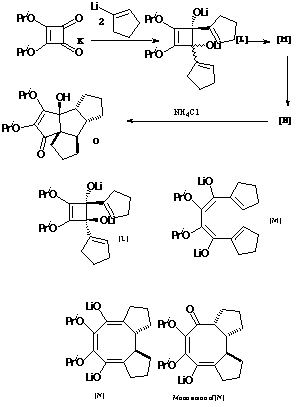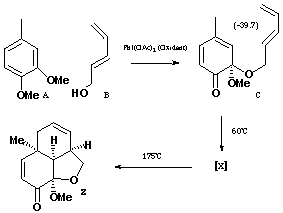
To use these models, move the mouse cursor to the model and press the mouse button (rhs button for a windows machine). From the menu that appears, select File/Save molecule as, and save the model to your local hard disk. Select a modelling program (e.g. Weblab Viewer/Medchem Explorer, CAChe, Chem3D, MacMolPlt/GAMESS), open the saved file, and then proceed with the desired calculation.