The possibility of carbon-carbon double bond formation within a ß-lactam ring
is suggested by the known tendency of penicillins with electron-withdrawing
substituents, such as phthalimido, at the 6-position to undergo base-promoted
rearrangement to thiazetinones, [6] a reaction for which the azetone 11
is reasonabily proposed as an intermediate. [7]
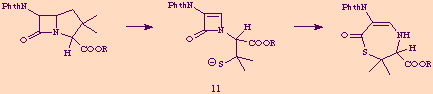
Moreover, a 3,4-unsaturaded ß-lactam has been detected spectroscopically, [8]
and azetinones fused to benzene or pyridine rings are well-known compounds.
[9]


