Professor Donald Craig
Research
Natural products synthesis | Novel pericyclic processes | De novo synthesis of highly subsituted aromatics
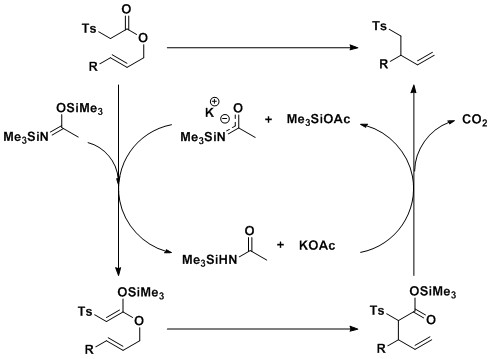
Recent
work in the group has shown that exposure of allylic tosylacetates to
sub-stoichiometric quantities of weak base (potassium acetate) and
silylating agent (N,O-bis(trimethylsilyl-acetamide): BSA) causes
in-situ silyl ketene acetal formation, [3,3]-sigmatropic rearrangement
and decarboxylation; these transformations provide homoallylic sulfones
in high yield in an overall decarboxylative Claisen rearrangement (dCr)
reaction.
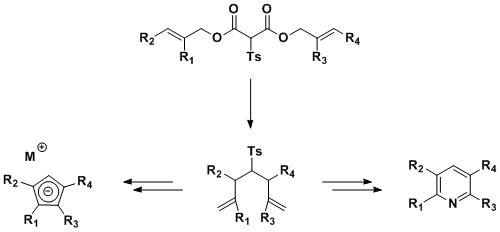
Doubly
allylic tosylmalonates can undergo tandem dCr reactions, where the
first rearrangement takes place at ambient temperature, while the
second frequently requires microwave irradiation. These
heptadiene products can be further elaborated to give access to densely
functionalised aromatic systems.
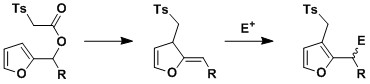
Incorporation
of the allylic moiety into a heteroaromatic ring leads to a
dearomatising dCr. Previous work within the group has shown that,
in certain cases, the dearomatised product can be isolated and further
synthetically manipulated, providing a facile route to 3-substituted
furans from furfural derived species.
Current
invesigations include the expansion of the above chemistry to
synthesise a range of cyclopentadiene-containing compounds, including
oligomeric and polymeric materials. An enantoioselective variant
of the dearomatising dCr is being developed also.
Stereospecific ring opening of aziridines
We
have previously shown that the ring opening of protected aziridines
with sulfone stabilised carbanions , can give access to highly
substituted piperidines and piperidones, structures commonly found in a
vast range of biologically active compounds.
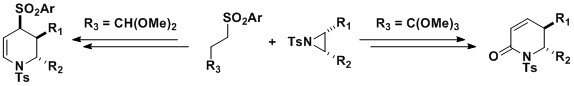
We
are currently exploring the reactivity of these piperidines towards
nucleophilic and electrophilic attack and the application of this
chemistry towards the synthesis of biologically important natural
products.
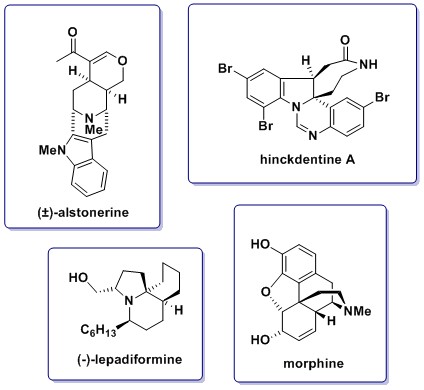
Natural Product Synthesis
The group maintains a strong interest in the field of natural product synthesis, utilising methodology developed within the group. Our recent synthesis of alstonerine included a regioselective, stereospecific ring opening of a trisubstituted aziridine; intramolecular Michael reaction, and a late-stage one-pot reduction-Pictet-Spengler cyclisation to give the polycyclic core. Current targets include lepadiformine, which has been shown to exhibit cytotoxic activity against cancer cell lines, and the indole containing alkaloid hinckdentine A.