
Adrenaline is a natural stimulant made in the adrenal gland of the kidney. Its biological name is epinephrine, from the Greek nephros for kidney. Adrenaline is carried in the bloodstream and affects the autonomous nervous system, which controls functions such as the heart rate, dilation of the pupils, and secretion of sweat and saliva.
Adrenaline is the body's activator, and is released in response to anxiety, exercise, or fear. This is the basis of the so-called 'fight-or-flight' reaction. When an animal is threatened, the options are usually either to stand its ground and fight, or run away as fast as possible. Both responses would require extra supplies of blood and oxygen in the muscles. Fright causes the brain to send signals to the renal glands which start pumping large amounts of adrenalin into the bloodstream. This increases the heart and breathing rate in preparation for the ensuing action.
 | Adrenaline in action. A cheetah running to catch its prey. |
Norepiniphrine (also called noradrenaline)is a molecule made in the brain and limbic system, and involves only a slight modification from the basic adrenaline structure. This molecule, however, serves a much different purpose. It is one of the main neurotransmitters, which means it is the chemical which jumps the gap (synapse) between nerve endings, so transmitting the signal between one nerve and the next. When it is working inside the brain, it gives rise to our thought processes and emotions. Another one of its main functions in the body is to maintain muscle tone in the blood vessels, thereby controlling blood pressure. People who suffer from hypertension (high blood pressure) are often given a drug called reserpine which works by greatly reducing the amount of norepinephrine in the nerve endings.
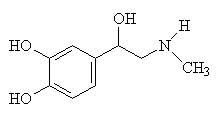 | 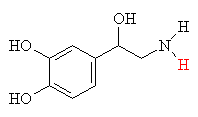 |
| Adrenaline (epiniphrine) | Norepiniphrine - the only change from adrenaline is a -CH3 has been replaced by an H, shown in red. |
The basic structure of norepiniphrine can be altered to produce a variety of drugs which act on the nervous system to either slow down or speed up certain bodily systems, or to make selected muscles contract or relax. This is particularly true for drugs used to control the heart rate (e.g. isoprenaline) or constriction of bronchi in the lungs (e.g. salbutamol).
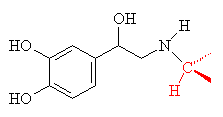 |  |
| Isoprenaline is used to increase heart rate in cases of bradycardia (slow heartbeat). The changes from norepiniphrine are just a replacement of H with -CH(CH3)2, shown in red. | Salbutamol is used to relax the bronchi in cases of asthma and is the drug used in some 'inhalers'. It is marketed under the name Ventolin. The changes from isoprenaline are shown in blue. |
Amphetamines are synthetic chemicals based upon a structure closely resembling that of adrenaline and norepiniphrine. These chemicals, therefore, can induce similar biological responses, such as acting as a stimulant, and creating a greater alertness and a feeling of prowess. The model for these chemicals, amphetamine, which was once widely available under the tradename Benzedrine is very similar to norepiniphrine, except that all the -OH groups are missing and an additional methyl has been added to the sidechain.
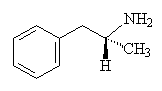 |  |
| Amphetamine (Benzedrine) | Dexedrine - the mirror image of Benzedrine |
Benzedrine was first used in the 1930s for the treatment of asthma, in the form of Benzedrine inhalers, and later other amphetamines were used as stimulants for armed forces in times of war (particularly WW2 and Vietnam), to enable soldiers to stay awake and alert for long periods. However, it was soon discovered that Benzedrine was a euphoriant, that is, it creates a feeling of well-being and buoyancy. Drug users quickly found a new way to get a 'high', and variants of benzedrine first hit the streets in the 1940s. Benzedrine is actually the name given to left-handed mirror image of the amphetamine molecule, but the corresponding right-handed molecule, called Dexedrine, is much more potent. A mixture of the two types (in the form of amphetamine sulphate) is commonly known as 'speed'.
Another euphoria-producing variation is methylamphetamine, or Methedrine, which differs from amphetamine by having a -CH3 group in place of one of the hydrogens on the -NH2.
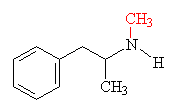 |  |
| Methedrine - the methyl group added to the amphetamine structure is shown in red | MDMA (Ecstasy) The methylenedioxy- group is shown in red |
A version of methedrine, 3,4-methylenedioxymethamphetamine (MDMA), or Ecstasy, has recently become notorious as the euphoric drug sold at 'acid-house' parties and as part of the 'rave scene'. Its stimulant effect allows the user to keep dancing for very long periods (at the severe risk of dehydration), and it also seems to force the brain into a mode whereupon it causes the body to continually repeat the same set of mechanical, rhythmic actions - which is why it is used as a supplement to dancing. But as with nearly all the amphetamines, long term use leads to dangerous medical problems, and sometimes even death.