 |
Chlorophyll is a green compound found in leaves and green stems of plants. Initially, it was assumed that chlorophyll was a single compound but in 1864 Stokes showed by spectroscopy that chlorophyll was a mixture. If dried leaves are powdered and digested with ethanol, after concentration of the solvent, 'crystalline' chlorophyll is obtained, but if ether or aqueous acetone is used instead of ethanol, the product is 'amorphous' chlorophyll.
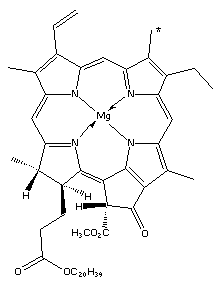
In 1912, Willstatter et al. (1) showed that chlorophyll was a mixture of two compounds, chlorophyll-a and chlorophyll-b:
 |
The two components were separated by shaking a light petroleum solution of chlorophyll with aqueous methanol: chlorophyll-a remains in the light petroleum but chlorophyll-b is transferred into the aqueous methanol. Cholorophyll-a is a bluish-black solid and cholorophyll-b is a dark green solid, both giving a green solution in organic solutions. In natural chlorophyll there is a ratio of 3 to 1 (of a to b) of the two components.
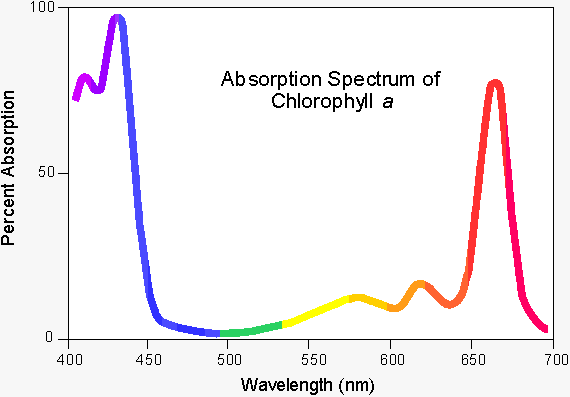
The intense green colour of chlorophyll is due to its strong absorbencies in the red and blue regions of the spectrum, shown in fig. 1. (2) Because of these absorbencies the light it reflects and transmits appears green.
Due to the green colour of chlorophyll, it has many uses as dyes and pigments. It is used in colouring soaps, oils, waxes and confectionary.
Chlorophyll's most important use, however, is in nature, in photosynthesis. It is capable of channelling the energy of sunlight into chemical energy through the process of photosynthesis. In this process the energy absorbed by chlorophyll transforms carbon dioxide and water into carbohydrates and oxygen:
 (CH2O) + O2
(CH2O) + O2The chemical energy stored by photosynthesis in carbohydrates drives biochemical reactions in nearly all living organisms.
In the photosynthetic reaction electrons are transferred from water to carbon dioxide, that is carbon dioxide is reduced by water. Chlorophyll assists this transfer as when chlorophyll absorbs light energy, an electron in chlorophyll is excited from a lower energy state to a higher energy state. In this higher energy state, this electron is more readily transferred to another molecule. This starts a chain of electron-transfer steps, which ends with an electron being transferred to carbon dioxide. Meanwhile, the chlorophyll which gave up an electron can accept an electron from another molecule. This is the end of a process which starts with the removal of an electron from water. Thus, chlorophyll is at the centre of the photosynthetic oxidation-reduction reaction between carbon dioxide and water.
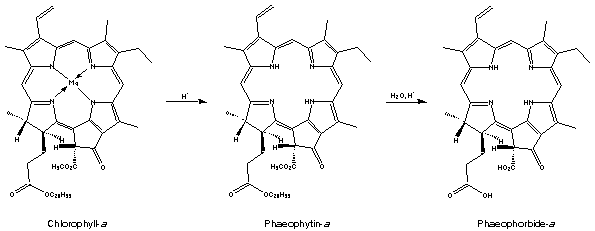
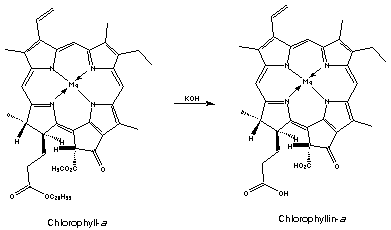
Treatment of cholorophyll-a with acid removes the magnesium ion replacing it with two hydrogen atoms giving an olive-brown solid, phaeophytin-a. Hydrolysis of this (reverse of esterification) splits off phytol and gives phaeophorbide-a. Similar compounds are obtained if chlorophyll-b is used.
Chlorophyll can also be reacted with a base which yields a series of phyllins, magnesium porphyrin compounds. Treatment of phyllins with acid gives porphyrins.
In plants chlorophyll is associated with specific proteins, for example, chlorophyll-a binding proteins are referred to as CP I, CP 47 and CP 43. With improving biochemical techniques for use on the membrane systems there has been an ever increasing success in the isolation and characterisation of these proteins.
Initially, detergents are used to break down the membrane into fragments, and these fragments are further broken down by the use of different detergents. These detergents work by replacing the membrane lipids which surround integral membrane proteins. The resulting particles are separated by polyacrylamide gel electrophoresis (a standard biochemical method) in the presence of sufficient detergent to keep them 'solubilised'. The activity and polypeptide composition can then be assayed as the particle is purified. The detergents work by substituting lipids at different spots in the membrane, this is also affected by the concentration of the detergent. One such detergent that is very commonly used is SDS-PAGE (sodium dodecyl sulfate-polyacrylamide). This is generally used as it has several advantages over other detergents: the separation can be carried out fairly rapidly and it also gives a good overall picture of the distribution of chlorophyll.
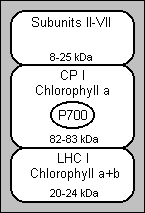
This figure shows a schematic representation of the major subfractions that can be isolated from thylakoid membranes. In PS I (photosystem I) an initial solubilisation produces large particles (called PS I-110). These particles contain two chlorophyll-protein complexes: the reaction centre chlorophyll-a protein (CP I) and a chlorophyll a+b complex (LHC I, light-harvesting complex) (3). PS I-110 also contains 6 to 8 polypeptides of lower molecular weight (8 to 25kDa, where 1 dalton=1 a.m.u.) that do not bind to chlorophyll, called Subunits II-VII. CP I, the reaction centre P700 chlorophyll-a protein, can be isolated from any of these mixtures by treatment with SDS (sodium dodecyl sulfate) or LiDS (lithium dodecyl sulfate) followed by electrophoresis.
Initial experiments done by Ogawa et al.(4) and Thornber (5) isolated two complexes by SDS-PAGE from SDS-solubilised membranes. One of these complexes, CP I, had a high apparent molecular weight and contained only chlorophyll-a. CP I is the most stable of the complexes and retained the photochemical activity of P700, the reaction centre of chlorophyll in PS I. It has a chlorophyll to P700 ratio of ~45 (6, 7, 8, 9, 10) and a beta-carotene to P700 ratio of ~8.
The nature of the reaction centre of chlorophyll, P700, is still unknown, as there is conflicting evidence. It has been suggested that this could be explained if there is a pair of electronically interacting chlorophyll-a molecules in the ground (reduced) state (P700), and that the unpaired electron of the P700+ (oxidised) state is localised on only one of the chlorophyll's (11). The other 40 to 50 chlorophyll-a molecules of CP I act as antennas, and are thought to be responsible for the 721nm fluorescence emission maximum (12, 13).
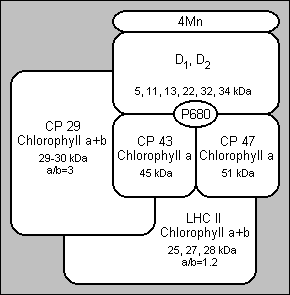
Improved extraction procedures gave oxygen evolving PS II (photosystem II) particles (BBY's). These particles are large pieces of granal membranes, probably lipid depleted (14, 15, 16, 17). Other detergent treatments have been employed to isolate the core particles from PS II. These core particles contain two reaction chlorophyll-a proteins, CP 47 and CP 43 and several non chlorophyll binding polypeptides (D1, D2), but are free from chlorophyll a+b complexes. Core particles which retain manganese have been successfully isolated with the two chlorophyll-a proteins and a limited number of other polypeptides. CP 47 and CP 43 can be purified from the other components of PS II by the use of SDS-PAGE or HPLC (high performance liquid chromatography) but they have no photochemical activity in isolation.
In 1977, a minor chlorophyll-a complex was detected by SDS-PAGE. The complex was rather unstable a contained a much lower percentage chlorophyll than CP I and was named CPa. It was then discovered that CPa was really two complexes: by solubilising thylakoid membranes with octyl gluside (a detergent), Camm and Green (18, 19) demonstrated the presence of the two complexes. These complexes are now named CP 47 and CP 43.
The PS II reaction centre is significantly more complex than the reaction centre of PS I, where P700 is clearly localised on the green complex CP I. P680, the reaction centre chlorophyll of PS II, is difficult to determine because the P680+ Pheo- charge separation decays within a nanosecond. P680 is currently considered to be a chlorophyll-a dimer, at least in the ground state.
Chlorophyll is essentially two parts: a substituted porphyrin ring and phytol (the long carbon chain). The porphyrin ring is an excellent chelating ligand, with the four nitrogen atoms binding strongly to a co-ordinated metal atom in a square planar arrangement. There are many examples of this including heme and vitamin B12.
Heme consists of a porphyrin similar to that in chlorophyll but with an iron(II) ion in the centre of the porphyrin. Heme is bright red. In the red blood cells of vertebrates, heme is bound to proteins forming hemoglobin. Hemoglobin combines with oxygen in the lungs, gills, or other respiratory surfaces and releases it in the tissues. In muscle cells, myoglobin, the name given to hemoglobin in muscles, stores oxygen as an electron source for energy-releasing oxidation-reduction reactions.
Vitamin B12 contains a cobalt ion at the centre of the porphyrin. Like heme, vitamin B12 is bright red. It is essential to digestion and nutritional absorption in animals.
Some other examples of porphyrin derivatives are shown below.
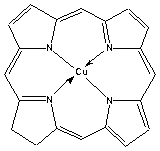
Another example is a co-ordinated iron species in which the porphyrin ring is also substituted: