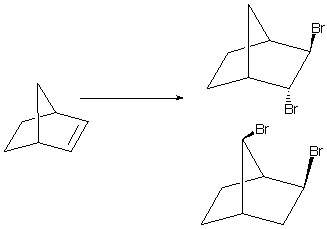

Both Br and C+ in this context can form bridging, so called non-classical species, as well as more conventional carbonium ions. Four isomers are therefore possible, two of which are conventional bromo carbonium ions (1,2) one is the bridging bromonium ion (3) and the fourth is a bridging (non-classical) carbonium ion (4). These non-classical species had a fascinating and very controversial history during the 1950s to 1970s, some people arguing that they did not exist, and others that it did. For years, a "definitive experiment" to distinguish whether the norbornyl cation was classical or non-classical was sought. The argument was in the event actually settled by spectroscopic rather than chemical experiments (photoelectron and 13C).
| 1. Classical carbocation |
|---|
At the MP2/6-31G* ab initio level, the non-classical carbonium ion/classical carbonium bridging Br structures are the ONLY structures that can be found. The "classical" structures do not appear to exist on the potential surface, in direct contrast to the PM3 result. At this level of theory, the C bridging isomer is essentially the same in energy as the Br brdging isomer. Opening of the former would lead to 1,3 dibromination with syn di-bromo stereochemistry, opening of the latter would lead to anti Br stereochemistry. Thus the two products observed in this reaction arise from two different non-classical intermediates of essentially similar energy.
Its noteworthy that the same effect is seen for reaction between norbornene and the electrophiles Cl2 and ICl. I2 however gives an anti-1,3 product, whilst F2 (actually XeF2) gives a syn 1,2 product, maybe by direct quenching of the non-classical carbonium ion, which becomes possible because the F is much smaller sterically than the Br.