


.

1,2,5-Trideoxy-2-fluoro-1,5-imino-D-glucitol, the 2-deoxyfluoro derivative of 1-deoxynojirimycin as well as 1,2,5-trideoxy-1-fluoro-2,5-imino-D-mannitol, a five-membered ring analogue, were synthesized and their glucosidase inhibitory activities compared with data from the respective parent compounds and other structurally related molecules.
.

Five- and six-membered ring sugar analogues bearing a basic nitrogen instead of oxygen in their ring systems have been demonstrated to be very interesting classes of natural as well as non-natural products. Many representatives exhibit significant biological properties of potential medicinal interest, such as (amongst others) anti-diabetic and anti-retroviral activities, due to their strong interference with glycosidases of various sources.1 Certainly one of the most well known representatives is the isosteric analogue of D-glucopyranose, 1-deoxynojirimycin (1,5-dideoxy-1,5-imino-D-glucitol, 1), which had been synthesised by Paulsen and coworkers 2 long before it was shown to be a natural product 3 and a powerful reversible inhibitor of a wide range of alpha- as well as beta-glucosidases.1 A virtually isosteric analogue, the five-membered ring 2,5-dideoxy-2,5-imino-D-mannitol (2), also a natural product , exhibits the same or even more pronounced biological activity against a range of glucosidases.1,5
| Molecule | # | R | R1 | R2 | R3 |
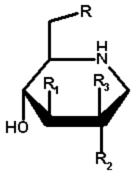 |
1 | OH | OH | OH | H |
| 3 | OH | OH | H | H | |
| 5 | OH | OH | NHAc | H | |
| 7 | OH | OH | H | OH | |
| 8 | OH | OH | H | OEt | |
| 9 | OH | OH | H | F | |
| 10 | H | OH | OH | H | |
| 12 | F | OH | OH | H | |
| 13 | OH | H | OH | H | |
| 14 | OH | OMe | OH | H | |
| 15 | OH | F | OH | H | |
| 16 | OH | OH | F | H | |
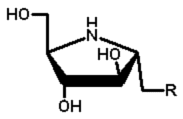 |
2 | OH | |||
| 4 | H | ||||
| 6 | NHAc | ||||
| 18 | F | ||||
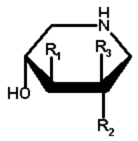 |
11 | OH | OH | H | |
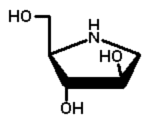 |
17 |
Several natural as well as non-natural derivatives of compounds 1 and 2 bearing various modifications around the ring system are known to date. The 2-deoxy derivative of inhibitor 1, the natural product fagomine (3), was reported to exhibit only very limited enzyme inhibitory activity. 1 The 1-deoxy derivative of compound 2, 1,2,5-trideoxy-2,5-imino-D-mannitol (4), also a natural product, is, interestingly, a powerful inhibitor of beta-mannosidases rather than a glucosidase inhibitor. The synthetic 2-acetamido-2-deoxy derivative of 1, compound 5, is a very powerful inhibitor of N-acetylhexosaminidases 1 while its 5-membered ring analogue 6, still being very active, shows activity reduced by a factor of fifteen when compared to 5. 7b
| Superposition 1 | ||
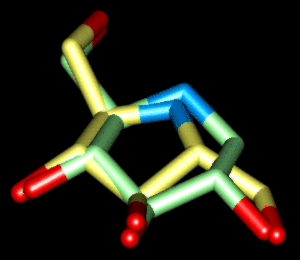 |
||
| green | Comp. 1 (6-memb. ring) | PDB-File |
| yellow | Comp. 2 (5-memb. ring) | PDB-File |
| Superposition 2 | ||
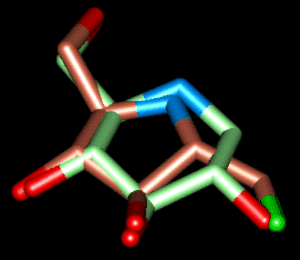 |
||
| green | Comp. 1 (6-memb. ring) | PDB-File |
| brown | Comp. 18 (5-memb. ring) | PDB-File |
| Superposition 3 | ||
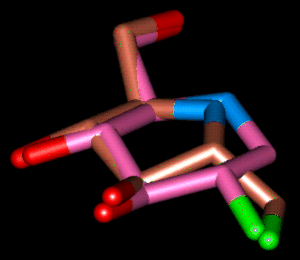 |
||
| pink | Comp. 16 (6-memb. ring) | PDB-File |
| brown | Comp. 16 (5-memb. ring) | PDB-File |
The naturally occurring C-2-epimer of 1, 1,5-dideoxy-1,5-imino-D-mannitol (7) is an inhibitor of mannosidases from various sources whereas the recently synthesized 2-O-ethyl- and 2-deoxy-2-fluoro analogues, 8 and 9, do not exhibit reasonable activities against jack bean alpha-mannosidase or beta-glucosidase from almonds, indicating the importance of the hydroxy function at C-2 for recognition/inhibitory activity with these enzymes.
In context with a programme aiming at evaluating the relative significances of the hydroxy functions in compounds 1 and 2 for glucosidase inhibitory activity we have synthesized analogues by replacing alcohol groups with less polar substituents such as fluorine atoms and compared their activities to the parent compounds as well as other known derivatives.
Modifications in the side chain of 1-deoxynojirimycin compare as follows: Replacing the hydroxymethyl group with a less polar methyl group by deoxygenation of C-6 (compound 10) reduced the activity against beta-glucosidase from almonds by a factor of 2 and against alpha-glucosidase from yeast by a factor of about 4. 7 Just as pronounced was the decline of activity upon formal removal of the side chain to give the meso-compound 1,5-dideoxy-1,5-imino-D-xylitol (11) which is practically devoid of glucosidase inhibitory power. 8 Interestingly, formal replacement of the hydroxymethyl group with a fluoromethyl substituent (compound 12) reduced the activity against beta-glucosidase from Aspergillus wentii by a factor of 200 while beta-glucosidase from almonds was inhibited with half the power of the parent compound. 9 The compound showed the same inhibition characteristics and strength with alpha-glucosidase from yeast but was a hundredfold less active with alpha-glucosidase from rice. These findings point to the fact that the hydroxyl group at C-6 is very important for binding but in case of some glucosidases can be replaced by other strongly electronegative substituents such as fluorine with only minor effects on the inhibitory power.
Modifications at C-4 of 1-deoxynojirimycin are scarce and so are alterations at C-3, such as deoxygenation (13), 10 O-methylation (14) 11 as well as the substitution of OH-3 by a fluorine atom (15), 12 in all cases destroying the inhibitory power of the molecule. 13 Position 2 is probably the most distinguishing for enzyme recognition and specificity in the respective inhibitor molecule. So it is not surprising that removal of OH-2 yields an inferior inhibitor as demonstrated by the lack of activity of fagomine (3) as mentioned before. For the same reason large substituents can also be expected to be deletorious to the activity (compare compound 8).
.

| Scheme 1: Synthesis of Compound 16 | |
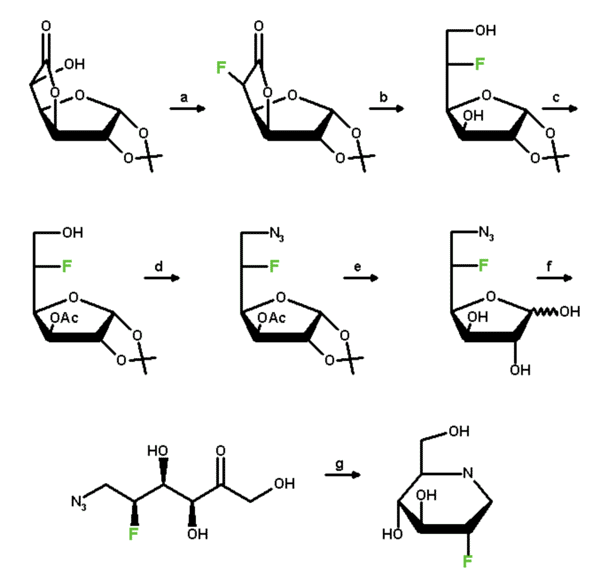 |
|
| a | Tf2O, Pyr.;Bu4NF; CH3CN |
| b | NaBH4/MeOH |
| c | TritCl, Pyr, Ac2O; BF3.Et2O/MeOH |
| d | Tf2O/Pyr.; NaN3/DMF |
| e | NaOMe, MeOH; IR 120 [H+], CH3CN/H2O |
| f | D-xylose isomerase (sweetzymeT) |
| g | H2, Pd/C |
Much to our own surprise we have recently found that the 2-deoxyfluoro derivative of 1-deoxynojirimycin, 16, despite being inactive against beta-glucosidase from almonds and hundred times less active against beta-glucosidase from Asp. wentii than the parent compound, exhibited interesting activity against alpha-glucosidase from yeast, at least comparable with the inhibitory power of 1-deoxynojirimycin itself. It seems noteworthy, though, that the equilibrium is reached after distinctly longer periods of time than usually measured with this class of inhibitors.
In the light of this result it seemed to be interesting to have a look at derivatives of 2,5-dideoxy-2,5-imino-D-mannitol (2). This compound is a powerful glucosidase inhibitor even superior to 1-deoxynojirimycin (1). This high activity was attributed to the fact that in the two molecules all hydroxyl groups as well as the ring nitrogen are superimposable, OH-1 of the five-membered ring 2 taking the place of OH-2 in compound 1.
Only three noteworthy derivatives of meso-compound 2 have been found in Nature or synthesised to date.
Formal removal of one of the hydroxymethyl side chains yields 1,4-dideoxy-1,4-imino-D-arabinitol (17), a natural product and nearly 20 times as active as compound 2 against alpha-glucosidase from yeast but practically inactive against almond beta-glucosidase. 14 Formal replacement of a hydroxymethyl by a methyl substituent gives also a natural product (4) which, remarkably, is rather a powerful inhibitor of beta-mannosidases than of glucosidases as mentioned before.
| Scheme 2 | |
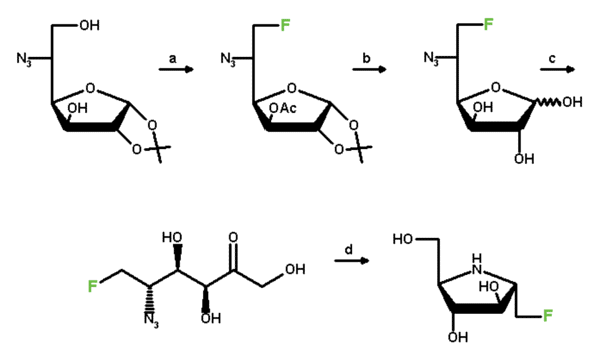 |
|
| a1 | TritCl/Pyr.; Ac2O/Pyr. CH2Cl2; BF3.Et2O/MeOH |
| a2 | Tf2O/Pyr.; Bu4NF/CH3CN |
| b | NaOMe, MeOH; IR 120 [H+], CH3CN/H2O |
| c | D-xylose isomerase (sweetzyme T) |
| d | H2, Pd/C |
We have recently synthesised the 1-deoxyfluoro analogue (18, Scheme 2) of compound 2 which was found to be by factors of 80 and 150 less active against alpha-glucosidase from yeast and beta-glucosidase from almonds, but about half as powerful as the parent compound against beta-glucosidase from Asp. wentii and invertase. Remarkably, compared with 1-deoxynojirimycin, compound 18 is still about half as active against yeast alpha-glucosidase and exhibits about 20% of the activity of the former against beta-glucosidase from almonds.
| Table: Glucosidase Inhibitory Activities of Compounds Ki-Values in Micromolar | ||||||
| Compound | Enzymes | |||||
| alpha-Glucosidases | beta-Glucosidases | |||||
| Yeast | Rice | Almonds | Asp. wentii | Agrobact. faecalis | Invertase Yeast | |
| 6-membered rings | ||||||
| 1 | 25 (pH 6) |
0.015 (pH 6) |
48 (pH 5) |
0.3 (pH 5) |
X | - |
| 3 | >1000 | - | - | - | - | - |
| 9 | - | - | >1000 7 |
- | - | - |
| 10 | 1560 7 |
- | 780 7 |
- | - | - |
| 11 | - | - | 430 8 |
- | - | - |
| 12 | 29 (pH 6) |
8 (pH 6) |
600 9 |
380 9 |
- | - |
| 14 | 1200 (pH 6) |
0.20 (pH 6) |
>20000 (pH 6) |
350 (pH 5) |
- | - |
| 15 | 750 | - | >5000 | >5000 | - | - |
| 16 | ca. 4 (pH 6) |
- | 2000 (pH 4.5) |
36 (pH 4.5) |
X | - |
| 5-membered rings | ||||||
| 2 | 0.73 (pH 6) |
- | 1.7 (pH 6) |
57 (pH 4.5) |
X | 3.5 (pH 6) |
| 17 | 0.18 14 |
- | 200 14 |
- | - | - |
| 18 | 57 (pH 6) |
- | 260 (pH 6) |
190 (pH 5) |
30 | 8.5 (pH 6) |
In the light of the unexpectedly high activities of deoxyfluoro compounds 16 and 18, examination of other derivatives bearing polar/electronegative substituents at the respective carbons might be interesting.
.

General methods: Melting points were recorded on a Tottoli apparatus and are uncorrected. Optical rotations were measured on a JASCO Digital Polarimeter with a path length of 10 cm. NMR spectra were recorded at 200 and 300 MHz, respectively (1H) and 50.29 and 75.47 MHz, respectively (13C) in CDCl3 for protected compounds and in D2O or methanol-d4 for free inhibitors. Chemical shifts are given in delta values. The signals of the protecting groups are in the expected regions and not listed explicitly. TLC was performed on precoated aluminium sheets (Merck 5554). For column chromatography Silica Gel 60 (Merck) was used.
5-Deoxy-5-fluoro-1,2-O-isopropylidene-beta-L-idofuranose was prepared according to a published procedure (15).
3-O-Acetyl-6-azido-5,6-dideoxy-5-fluoro-1,2-O-isopropylidene-beta-L-idofuranose.- To a 5% solution of 5-Deoxy-5-fluoro-1,2-O-isopropylidene-beta-L-idofuranose in dichloromethane containing 10% pyridine, 4-bromobenzenesulfonylchloride (1.2 equiv.) was added and the mixture kept at 10 �C until tlc indicated that all starting material had been consumed. Acetic anhydride (1.5 equiv.) was added and the mixture was kept at ambient temp. overnight. Methanol (3 equiv.) was added and the mixture was consecutively washed with 5% aqueous HCl and 5% aqueous sodium bicarbonate. After drying (sodium sulfate) and filtration, the filtrate was concentrated under reduced pressure. To a 5% solution of the residue in N,N-Dimethylformamide, sodium azide (10 equiv.) was added and the mixture was stirred at 40 �C until tlc indicated the quantitative formation of a new compound. Dichloromethane was added, the precipitate was filtered off and the filtrate was concentrated under reduced pressure. Chromatography on silica gel yielded compound as a colourless syrup. It had [alpha]20,D -29 (c 2.7, ethyl acetate). 13C-NMR (delta in ppm): 104.7 (C-1), 90.0 (JC,F 185 Hz, C-5), 83.5 (C-2), 77.7 (JC,F 2.4 Hz, C-3), 77.2 (JC,F 11 Hz, C-4), 51.7 (JC,F 23.1 Hz, C-6); 1H-NMR: 5.97 (d, 1 H, J1,2 3.7 Hz, H-1), 5.25 (d, 1 H, J3,4 3.3 Hz, H-2), 4.80 (dddd, 1 H, J4,5 5.1 Hz, J5,6 6.2 Hz, J5,6' 4 Hz, J5,F 47.8 Hz, H-5), 4.55 (ddd, 1 H, J2,3 0.7 Hz, J2,F 1.8 Hz, H-2), 4.36 (ddd, 1 H, J4,F 19.5 Hz, H-4), 3.56 (ddd, 1 H, J6,6' 13.7 Hz, J6,F 21.4 Hz, H-6), 3.45 (ddd, 1 H, J6'F 24.4 Hz, H-6').
For analytical purposes, a sample was de-O-acetylated employing standard Zemplen conditions to give, after chromatographic purification, 6-Azido-5,6-dideoxy-5-fluoro-1,2-O-isopropylidene-beta-L-idofuranose as colourless crystals (mp. 110-111 �C). It had [alpha]20,D -66 (c 1.20, ethyl acetate). 13C-NMR: 104.9 (C-1), 91.5 (J5,F 176 Hz, C-5), 85,4 (C-2), 79.4 (J4,F 18.3 Hz, C-4), 75.2 (J3,F 6.7 Hz, C-3), 51.4 (J6,F 22.1 Hz, H-6); 1H-NMR: 5.99 (d, J1,2 3.7 Hz, H-1), 4.90 (dddd, 1 H, J4,5 6.5 Hz, J5,6 4.6 Hz, J5,6' 4.6 Hz, J5,F 48.5 Hz, H-5), 4.51 (dd, 1 H, H-2), 4.30 (ddd, J4,F 15 Hz, H-4), 4.25 (broad signal, 1 H, H-3), 3.63 (dd, 2 H, J6,F = J6'F 23.5 Hz, H-6,6'), 2.47 (broad signal, 1 H, 3-OH).
6-Azido-5,6-dideoxy-5-fluoro-alpha/beta-L-idofuranose.- Deprotection (16) of compound with the aid of acidic ion exchange resin Amberlite IR 120 in a mixture of acetonitrile/water 1:1 (v/v) at 50 �C furnished the free aldofuranose as a colourless syrup. 13C-NMR: 104.4, 97.5 (C-1 alpha/beta), 94.4, 94.2 (J5,F 175 Hz, C-5 alpha/beta), 82.0, 78.6 (J4,F 17.5 Hz, C-4 alpha/beta), 82.0, 77.7 (J2,F < 2 Hz, C-2 alpha/beta), 76.7, 76.6 (J3,F 6 Hz, C-3 alpha/beta), 52.9, 52.6 (J6,F 15 Hz, 21 Hz, C-6 alpha/beta).
6-Azido-5,6-dideoxy-5-fluoro-L-sorbose.- Following the reported procedure (16), 6-Azido-5,6-dideoxy-5-fluoro-alpha/beta-L-idofuranose was isomerised with the aid of Sweetzyme T (Novo) to yield, after chromatographic separation from the starting material, the open-chain L-sorbose derivative as a colourless syrup. It had [alpha]20,D -56 (c 1.7, methanol). 13C-NMR: 212.4 (C-2), 94.4 (J5,F 175.4 Hz, C-5), 76.6 (J3,F 5.4 Hz, C-3), 72.5 (J4,F 20.3 Hz, C-4), 67.8 (C-1), 52.6 (J6,F 21.3 Hz, C-6); 1H-NMR: 4.74 (dddd, 1 H, J4,5 5.8 Hz, J5,6 5.8 Hz, J5,6' 3.7 Hz, J5,F 48.4 Hz, H-5), 4.50 (dd, 2 H, J1,1' 18 Hz, H-1,1'), 4.30 (d, 1 H, J3,4 3.1 Hz, H-3), 4.12 (ddd, 1 H, J4,F 17 Hz, H-4), 3.66 (ddd, 1 H, J6,6' 14.3 Hz, J6,F 23.9 Hz, H-6), 3.57 (ddd, 1 H, J6'F 26.8 Hz, H-6').
1,2,5-Trideoxy-2-fluoro-1,5-imino-D-glucitol (1,2-dideoxy-2-fluoronojirimycin, 16).- Catalytic hydrogenation of 6-Azido-5,6-dideoxy-5-fluoro-L-sorbose in dry methanol over Pd/C (10%) at ambient temp. and atmospheric pressure gave the known 1-deoxynojirimycin derivative 16 as a white solid. After crysallization from methanol/ether, physical data matched reported values (17).
5-Azido-5,6-dideoxy-6-fluoro-alpha/beta-D-glucofuranose and 5-Azido-5,6-dideoxy-6-fluoro-D-fructose were synthesized following our previously reported procedures.(18, 19)
1,2,5-Trideoxy-1-fluoro-2,5-imino-D-mannitol (18).- Catalytic hydrogenation of 5-Azido-5,6-dideoxy-6-fluoro-D-fructose in dry methanol at ambient temperature and atmospheric pressure for 16 h furnished, after crystallization from methanol/ether, the title compound as white crystals. It had mp., [alpha]20,D 21.7 (c 0.7, methanol).
13C-NMR: 84.9 (J1,F 169 Hz, C-1), 79.2 (J4,F < 2 Hz, C-4), 78.7 (J3,F 6.6 Hz, C-3), 62.9 (C-6) 62.85 (J2,F 18.6 Hz, C-2); 1H-NMR: 4.51 (ddd, 1 H, J1,1' 9.7 Hz, J1,2 3.8 Hz, J1,F 37.6 Hz, H-1), 4.44 (ddd, 1 H, J1',2 5.7 Hz, J1'F 37.6 Hz, H-1'), 3.83 (m, 2 H, H-3, H-4), 3.69 (dd, 1 H, J5,6 4.1 Hz, J6,6' 11.3 Hz, H-6), 3.61 (dd, 1 H, J5,6' 5.3 Hz, H-6'), 3.17 (m, 1 H, J2,F 22 Hz, H-2), 2.97 (m, 1 H, H-5).
.

We appreciate financial support by the Austrian Fonds zur Förderung der Wissenschaftlichen Forschung, Vienna, (Projects 7335, 8415, and 10067) as well as NOVO, Denmark, for the generous gift of Sweetzyme T. We would like to thank Dr. Henry Rzepa, Imperial College London, for his great work in organizing ECTOC and his support during setup of our contribution.
.

.
