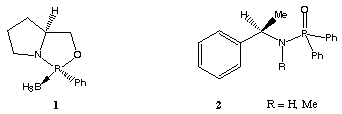
The reduction of unsymmetrical ketones to chiral alcohols using an asymmetric catalyst is an important reaction in organic synthesis[2]. Almost all of those catalysts fall into one of two major classes: 1) transition metal catalysts modified by chiral phosphines[3] or 2) oxazaborolidines developed by Itsuno and other authors[4]. The first class of catalyst furnish products of very high enantiomeric excess but are limited by the necessity for a nearby co-ordinating group in the substrate for optimum results. The second class of catalyst also give extremely high enantiomeric inductions but it is necessary that these reactions are prepared under conditions in which water is rigorously excluded. Some asymmetric ketone reduction catalysts do not belong to the above mentioned major classes.
The 1,3,2-oxazaphospholidine borane complex 1 furnished secondary alcohols of good enantiomeric excess[5,1e]. This compound has the advantage of easy preparation and high stability. Wills et al. recently reported that chiral phosphinamides such as 2 are efficient catalysts for the asymmetric borane reduction of ketones[6].

Scheme 1: novel classes of catalysts for the asymmetric reduction of ketones
In context with our studies on the utilization of the industrial waste material (all-R)-2-azabicyclo[3.3.0]octane-3-carboxylic acid benzyl ester we synthesized the enantiomeric pure (all-R)-2-azabicyclo[3.3.0]octane (all-R)-3 by a published procedure[7].
In this paper we describe the synthesis of the homochiral phosphinamides (all-R)-4 and (all-R)-5 from (all-R)-3.
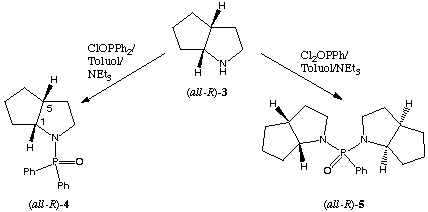
Scheme 2: synthesis of new chiral phosphinamides
(all-R)-4: The amine (all-R)-3 (1 eq) was dissolved in dry toluene, 1.5 equivalents of triethylamine was added and the solution was cooled with an ice-bath. Diphenylphosphinic chloride (1 eq) was slowly added and after completly addition the mixture was stirred at room temperature for 24 h. The triethylamine hydrochloride was removed by filtration. The toluene distilled off under reduced pressure. Pure (all-R)-4 was obtained from crude (all-R)-4 as a colourless solid (yield = 43%[8]) by recrystallisation from toluene.
(all-R)-5: Phenylphosphonic dichloride (0.5 eq) was added to a mechanically stirred and chilled solution of (all-R)-3 (1 eq) and triethylamine (1.5 eq) in dry toluene at such a rate as to keep the temperature below 5deg.C. After stirring for 24 h at room temperature the triethylamine hydrochloride was removed by filtration and the toluene by rotary evaporation. After standing for 24 h at +4deg.C a brown solid precipitated which was removed by filtration. The solvent was completly distilled off and (all-R)-5 could be obtained as a yellow oil (yield = 45%8). Both compounds are stable to air and moisture. Investigations of the catalytic ability and the asymmetric induction in reductions by borane are still in progress and our findings will be reported in due course.
Acknowledgement: We express our thanks to Hoechst AG and the Fonds der Chemischen Industrie for the generous providing with chemicals and for the financial support.