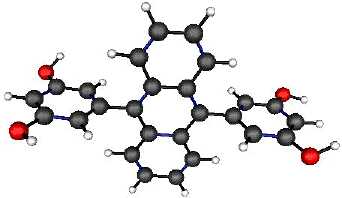
 The resorcinol based aromatic
molecule shown below forms hydrogen-bonded lattices in the solid state, the
cavities of which are large enough to accomodate three small organic
molecules, such as methyl methacrylate and cyclohexadiene. Click here to see a 3D
model of the cavity and the reagents trapped inside. Other types of
cavities formed from covalently bound porphyrinic-hosts (as opposed to
cavities formed via intermolecular hydrogen bonds) have been synthesised by
Jeremy Sanders at Cambridge. Calculated models for these can be seen
here.
The resorcinol based aromatic
molecule shown below forms hydrogen-bonded lattices in the solid state, the
cavities of which are large enough to accomodate three small organic
molecules, such as methyl methacrylate and cyclohexadiene. Click here to see a 3D
model of the cavity and the reagents trapped inside. Other types of
cavities formed from covalently bound porphyrinic-hosts (as opposed to
cavities formed via intermolecular hydrogen bonds) have been synthesised by
Jeremy Sanders at Cambridge. Calculated models for these can be seen
here.
For further information, see K. Endo, T. Koike, T. Sawaki, O. Hayashida,
H. Masuda and Y. Aoyama, J. Am. Chem. Soc., 1997, 119, 4117.
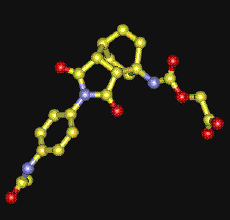 Pericyclic reactions are unusual
in that very few enzymes which catalyse such reactions are known.
Artificial catalytic antibodies ('abzymes') can be created which can
perform this feat, and a prominent recent example is the isolation and
structural determination of the so-called "Diels-Alderase". Here, the
Diels-Alder transition state was mimicked using an derivative of a
bicyclo-octane know as FRA. This acts as an inhibitor to the real
Diels-Alder reactions, and the enzyme can be crystallised out with FRA
trapped in the active site.
Pericyclic reactions are unusual
in that very few enzymes which catalyse such reactions are known.
Artificial catalytic antibodies ('abzymes') can be created which can
perform this feat, and a prominent recent example is the isolation and
structural determination of the so-called "Diels-Alderase". Here, the
Diels-Alder transition state was mimicked using an derivative of a
bicyclo-octane know as FRA. This acts as an inhibitor to the real
Diels-Alder reactions, and the enzyme can be crystallised out with FRA
trapped in the active site.
Another catalysed pericyclic reaction is the [3,3] sigmatropic type Claisen rearrangement of Chorismate to pre-phenate, a reaction induced by the enzyme Chorismate mutase for the Claisen rearrangement.
For further information on Diels-Alderase, see Katayama_K, Kobayashi_T,
Oikawa_H, Honma_M, Ichihara_A, Biochimica Et Biophysica Acta-Protein
Structure And Molecular Enzymology, 1998, Vol.1384, pp.387-395;
Heine_A, Stura_EA, YliKauhaluoma_JT, Gao_CS, Deng_QL, Beno_BR, Houk_KN,
Janda_KD, Wilson_IA, SCIENCE, 1998, 279, No.5358, pp.1934-1940.