 STRUCTURE ACTIVITY
RELATIONSHIP
STRUCTURE ACTIVITY
RELATIONSHIP
The structure activity relationships of taxol has been studied by
a whole range of researchers. Many derivatives have already been
synthesised and comparing their activities, has revealed which part
of the molecule is responsible for it's biological activity.
The Taxol molecule can be divided into two distinguishable parts:-
1. Side Chain
The side chain of taxol is critical for maintaining activity. It
seems to be a major site of recognition in the tubulin-Taxol binding
mechanism.
Studies by Portier and Swindell and other prominent scientists has
revealed the following features:-
- Protection of the C2' hydroxyl group as an ester, results in a
loss of activity in terms of microtubilin stabilisation, but not
in cytotoxicity.
- The derivative of the C2' hydroxyl has to be carried out with
water labile functions in order to release it into the body.
- The C3' bound nitrogen can be replaced by an oxygen atom
without loss of activity.
- The C3' amide-acyl group is critical in the molecules
bio-activity, but may be alkyl or aromatic in nature.
- The C3' aryl group is needed, because replacing it with a
methyl group leads to a reduction in activity by 19- fold.
- The 2'R, 3'S configuration can be changed to S,S or R,R (but
not S,R) without a major loss in activity.
2. Taxol Skeleton
Taxol is made up of several rings: a six membered double bond
containing (A) ring, an eight membered (B) ring, another six membered
(C) ring and a four membered (D) ring. Modification of the taxol
skeleton and studying it's activity has lead us to the following
features:-
- Removal of the C2, C7 and the C10 acetyl groups doesn't seem
to have any effect on the activity.
- Removing the C2-O-benzoyl group causes a drastic reduction in
activity.
- Changing the eight membered (B) ring to a seven membered ring
still provides the molecule with tubulin depolymerization
properties.
- The oxetane (B) ring is crucial in maintaining the molecules
activity and ring opening leads to a dramatic decrease in
biological activity.
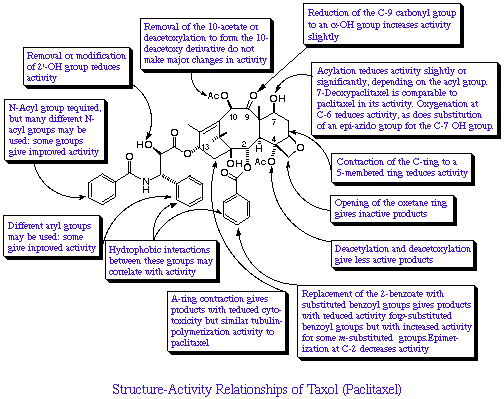
The following diagram summarises the structure-activity
relationship of Taxol.
REFERENCES
Einzig, A.I.; Hochster, H.; Wiernik, P.H.
Investigational New Drugs. 1991,
9, 59-64
Legha, S.S.; Benjamin, R.S. Cancer. 1990,
65, 2478-2481
 NEXT PAGE
NEXT PAGE

