| 1 | 2 | |
|---|---|---|
| 3 | 4 | |
| 5 | 6 | |
Co(acac)3 itself has three separate planar rings 1. Due to the octahedral coordination at the Co, these rings are not orthogonal to each other, and neither are the σ/π electrons, which consequently mix with each other. The resulting molecular orbital diagram is therefore quite complex and difficult to interpret. In order to retain σ/π separation, a simpler molecule with a plane of symmetry separating the σ/π electrons is modelled. Compound 3 is known to be planar2, although increasing the steric hindrance can in fact result in the two rings being orthogonal (4)3. Based on this precedent, Co(acac)F2 is modelled. This is predicted to exist as two isomers; the first 5 has all the atoms in a plane as with 3, the second 6 has one F atom in the plane, and the other above the plane.
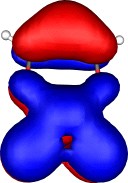
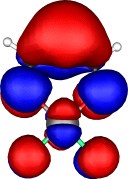
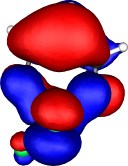
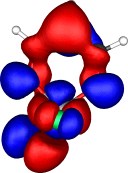
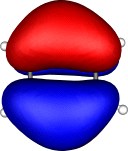
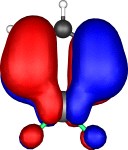
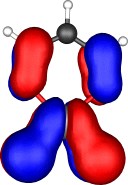
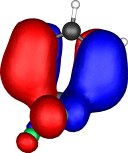
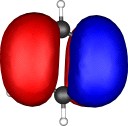
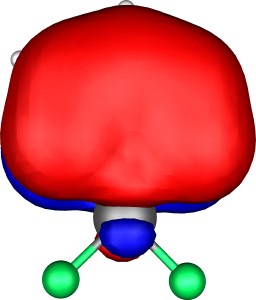
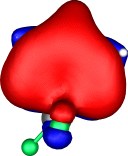
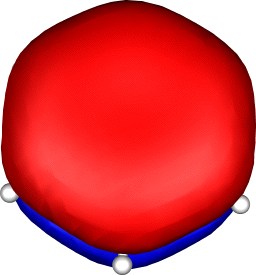
A total of five π Molecular orbitals can be discerned (B3LYP/6-31G(d) calculation for 5, with orbitals contoured at a threshold of 0.02 a.u. These can be further partitioned into three associated with the ring, in linear combination with the 2p AOs on each F atom. The three π-MOs can be associated with 6π (4n+2) aromaticity of the ligand ring. This analogy is made clearer by juxtaposing the 3 π MOs of benzene itself. The diagram for isomer 6 is essentially similar, suggesting the precie geometry at the metal does not qualitatively change the aromaticity of the metallacyclic ring.
| Molecular orbitals of 5 | Molecular orbitals of 6 | MOs of benzene | ||
|---|---|---|---|---|
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
 |
 |
 |
||
It is possible to measure the magnetic diatropic ring current by placing a so-called NICS probe at the centre of the ring. This will give a measure of the total magnetic diatropicity at this point generated by the ring current, but in this position it includes also a paratropic contribution from the σ electrons, as well as local magnetic effects due to proximity to the Co nucleus. To separate these latter contaminants, a NICS probe is better placed either 1A or 2A above this ring centroid (NICS(1) and NICS(2) respectively).
The NICS(1) and NICS(2) values for the Co(acac)F2 are respectively -33.6 and -15.2 ppm. The corresponding values for benzene itself are -12.8 and -5.5 ppm, which suggests that the C(acac) ring in 5generates a stronger diatropic ring current than benzene itself.
The two faces of the ring in the isomer 6 are no longer equivalent. Placing the NICS probe above the ring centroid on the same face as the axial F atom again generates local magnetic effects. Accordingly, the NICS(1) and NICS(2) probes are placed on the opposite face, well away from the F atom. The values are respectively -65.2 and -40.2 ppm. Whilst quantitatively, intrepreting these values may be difficult, they do appear to indicate a substantial diatropic ring current associated with the ring.