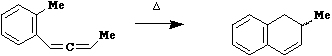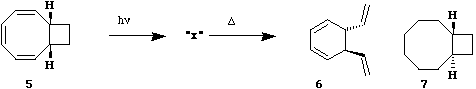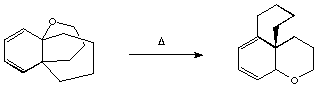Practice Problems in Pericyclic Reactions.
Copyright (c) 1994 - 1997 H. S. Rzepa
The following are a set of practice problems in Pericyclic Mechanisms, prepared
by Henry Rzepa for the
for a second year course at the Department of Chemistry,
Imperial College. Each of these problems is intended to
take between 1-3 hours to solve.
They are NOT typical of examination questions! Please note
also that the compound numbers are not consecutive between
questions.
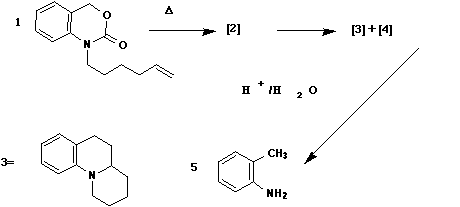
Qu. 1 When compound 1 is heated, a
transient intermediate 2 is formed, rapidly rearranging
to two new compounds 3 and 4. An aqueous workup of this
mixture, in which traces of acid were present, showed
that 4 had been converted to 5. Suggest structures for
compound 2-4, and mechanisms for all the steps in this
sequence.
Answer
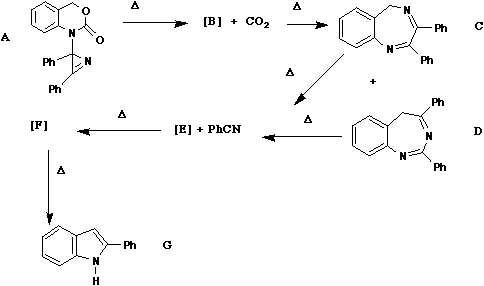
Qu. 2
When compound A is heated, carbon dioxide and
benzonitrile (PhCN) are eliminated and the final observed
product is the indole G. It is thought that the mechanism
of the reactions proceeds via a series of intermediates B
- F, the structures of two of which (D and E) are shown
below. Suggest probable structures for the other species
B, E and F and mechanisms for all the reactions. Pay
attention to why both C and D give the same intermediate
E, any selection rules that may relate to individual
steps and the nomenclature of each step.
Answer
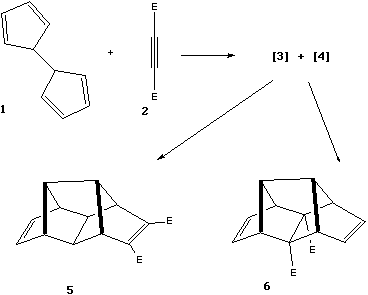
Qu. 3. a) When molecules 1 and
2 (E = CO2Me) are heated together, the transient
intermediates 3 and 4 are formed, with the eventual
isolation of 5 and 6 as the two final products. Propose
structures for 3 and 4 and a mechanism for the formation
of 5 and 6.
b) Propose
structures for A and B and mechanisms for the following
transformations.
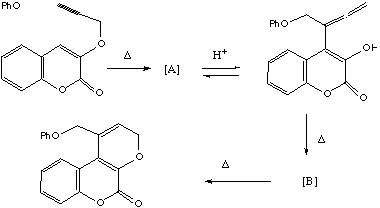
Answer
Qu. 4
Photolysis of compound 7 in benzene as solvent
produces the triene 8, which then decomposes thermally to
give butadiene and tetralin (12), via it is thought the
intermediates 9, 10 and 11. Propose and classify the
mechanisms for these transformations, and suggest a
structure for 11.
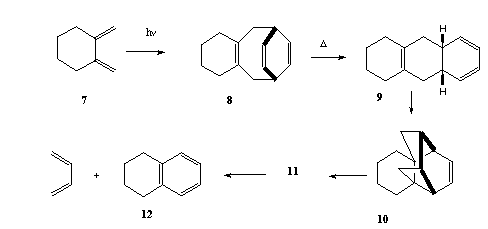
Answer
Qu.
5. a). Suggest a mechanism for the
following reaction which explains the observed
stereochemistry;
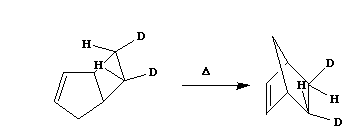
b).
Propose a structure for 1 consistent with the
spectral evidence and classify the type of pericyclic
reaction occuring, paying particular attention to the
expected stereochemistry of the product. 1H nmr includes
the following; d 1.43 (3H, s), 1.52 (3H, triplet, J
1.5Hz), 3.76 (1H, multiplet), 5.71 (1H, multiplet), 5.73
(1H, double doublet, J 10, 1.5Hz), 5.79 (1H, doublet, J
10Hz), 6.74 (1H) + phenyl protons. No carbonyl peak is
evident from the infra-red spectrum and no cyclopropyl
group is present in 1.
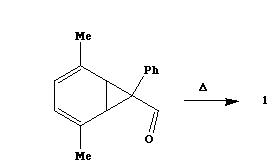
Answer
Qu. 6.
Tropylium tetraborate can be reduced with Zn/Hg to
ditropyl [A].
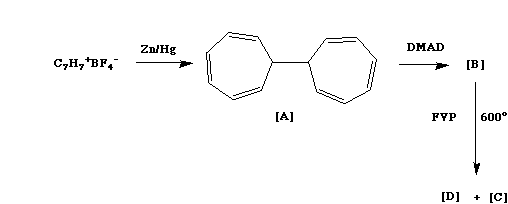
Ditropyl is in
equilibrium with an isomer, which when refluxed with an
excess of the dimethyl ester of acetylene dicarboxylic
acid (DMAD= MeO2C-CC-CO2Me) reacts to give a single
adduct [B] with the following nmr properties; d 6.0 (4H,
t, 4Hz), 4.05 (4H, broad singlet), 3.75 (12H, s), 1.2
(4H, double doublet, 3Hz, 2Hz), 0.9 (2H, triplet, 3Hz).
When [B] is subjected to flash vacuum pyrolysis at 600
(FVP) two compounds [C] and [D] are formed. Compound [D]
is stable and has the following nmr spectrum; d 7.62 (8H,
symmetrical multiplet), 3.90 (12H, s). The 1H nmr
spectrum for compound [C] includes the presence of
vinylic protons (4H), but on standing these slowly
disappear and a signal at d 7.37 (6H) due to benzene
takes their place. Suggest possible structures for [B] -
[D], paying particular attention to possible isomeric
forms, the mechanisms of their formation and their nmr
spectral properties. There is no need to propose a
mechanism for the conversion of [C] to benzene. Answer
Qu 7.
a) Propose a mechanism for the following
transformation, identifying any intermediates that might
be involved;
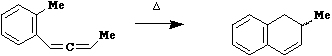
b) The
following transformation can take place via two
alternative pathways, one involving intermediates 2 and
3, the other involving a single intermediate 4. When 13C
label is introduced into the reactant, the product can be
shown to have the label distributed as shown. Suggest
possible structures for the intermediates 2-4, and on the
basis of the labelling information, indicate which
mechanistic pathway is actually followed.
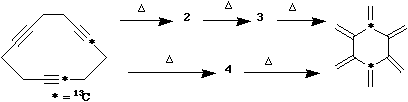
Answer
Qu 8. When compound 5 is photolysed in
heptane, a solution of "x" is formed. This solution shows
the following nmr properties. At -76� complex multiplets
in the 1H nmr spectrum are seen at δ 6.3-4.8 and at 3.3-
1.4 ppm. As the temperature is raised, both multiplets
are broadened and portions of each multiplet move
together and coalesce to a single multiplet at d 3.82 at
a temperature of 78�. The integral of this coalesced
multiplet is 1/6 of the total proton integral. On
recooling to -76�, the spectrum returns to its previous
appearance. If the solution is subjected to catalytic
hydrogenation, compound 7 is one of the products. If the
solution is heated to 98�, "x" then reacts to form a new
compound 6 in which the two vinyl groups are specifically
trans.
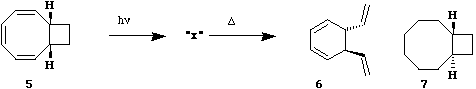
Suggest possible structure(s)
for the unknown "x", paying particular attention to
stereochemistry and the nmr behaviour. Suggest a
conformational explanation for why 6 has trans
stereochemistry (Hint: what must the most stable
conformation of the precursor to 6 be and how would you
estimate this quantitatively?) Answer
Qu. 9.
Classify the type of thermal reaction occuring in the
sequences below, indicating clearly any stereochemical
implications.
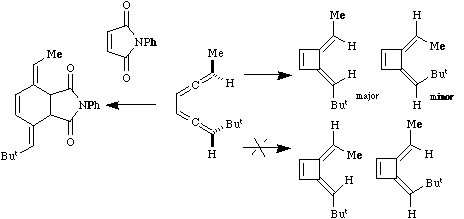
Answer
Qu. 10.
Classify the type of (thermal) pericyclic reaction
occuring for each of the eight steps shown below, showing
clearly how many electrons are involved in each step, and
discuss any obvious (or indeed less obvious!)
stereochemical implications and nomenclature.
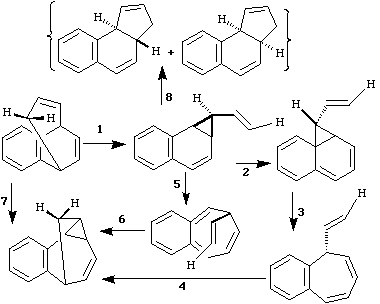
Answer
Qu 11. a) Propose a mechanism
for the following transformation, including an
explanation for the formation of two products and
indicating the number of electrons involved in each step.
Extrapolating from this explanation, suggest a related
third product that might have formed, but as it happens
was not detected with these particular substituents.
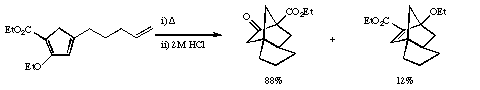
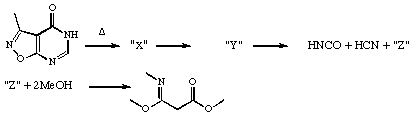
b) In the following
sequence, the technique of flash vacuum
pyrolysis results in the trapping of HNCO, HCN and
a novel third product Z on a cold finger. The species Z
shows at least one ir band in the region 2280 cm-1. When
trapped with two quivalents of methanol, Z gives the
product shown. Suggest possible identities for the two
intermediates �X and Y as well as Z, and mechanisms for
all the reactions, including details of nomenclature for
each step.
Answer
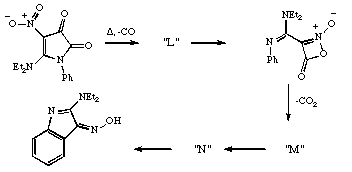
Qu 12.
In the following sequence, suggest possible structures
for the intermediates L, M and N, together with
mechanisms and nomenclature for their formation. Discuss
whether each step is formally allowed by the pericyclic
selection rules for thermal reactions, or whether it
requires the presence of traces of acid or base.
Answer
Qu 13. Suggest a nomenclature for the
following three substrates;
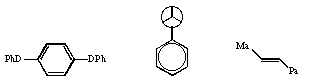
Answer
Qu 14. When a mixture of hexa-2,4-dienol, CS2, KOH and prop-2-ynyl
bromide is stirred for 12 hours at room temperature, the expected compound
1 is not isolated. Instead an oil "A" with the following spectroscopic
properties is obtained; [[nu]]max 1646 cm-1, dH 1.70 (3H, d, J 6Hz),
2.23 (1H, t, J 3Hz), 3.73 (2H, d, J 3Hz), 4.84 (1H, dd, J 7, 8 Hz), 5.16 (1H,
d, J 10 Hz), 5.27 (1H, d, J 17 Hz), 5.51 (1H, dd, J 8, 16.5 Hz), 5.72 (1H, dq,
J 6, 16.5 Hz), 5.88 (1H, ddd, J 7, 10, 17 Hz). On heating to 180*C,
compound 2 was isolated in 62% yield, via the presumed intermediate
shown. Identify compound "A", suggest mechanisms for the subsequent reactions
of 1, and assign the spectroscopic data, including a discussion of the
coupling constants. In compound 2, suggest a probable stereochemical
assignment for the methyl group marked with a
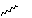
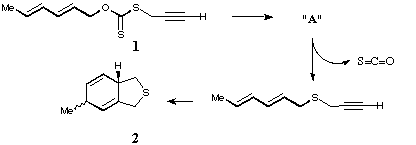
Answer
Qu 15. The 1,3 elimination of HCl from the imidoyl chloride isomers
shown proceeds rapidly at 0*C to give the two compounds 3 and
4. On standing at room temperature, these rearrange to "X" or "Y", for
which the following spectral data are obtained. X: dH 0.56 (3H, d, J 6.2 Hz),
2.40 (1H, dq, J 9.8, 6.2 Hz), 2.85 (1H, d, J 9.8 Hz), 8.51 (1H, s), 7.18-7.55
(9H, m); dC 5 (q), 15 (d), 33 (d), 54 (s), 155 (s) + aromatics. Y: dH 1.08 (3H,
d, J 6.2 Hz), 0.49 (1H, dq, J 5.5, 6.2 Hz), 2.66 (1H, d, J 5.5 Hz), 8.16 (1H,
s), 7.25-7.55 (9H, m); dC 28 (q), 14 (d), 32 (d), 58 (s), 153 (s) + aromatics.
On refluxing in benzene, either "X" or "Y" interconvert via the presumed
intermediacy of compound 5, which in turn slowly isomerises to 6,
the final isolated product. Suggest structures for "X" and "Y" based on
spectral and mechanistic evidence and propose likely mechanisms for their
formation and subsequent reactions, paying particular attention to the
stereochemistry of the species involved. (Hints: Try drawing alternative
resonance forms of 3 and 4, and pay particular regard to the
differences in the chemical shifts of X and Y. The multiplicities of the carbon
nmr signals are those obtained with off-resonance decoupling).
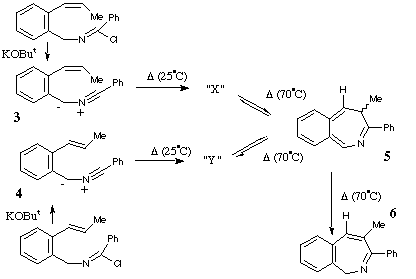
Answer
Qu 16. The synthesis of the naturally occuring coumarin ester
Gravelliferone is accomplished by heating the compound [1] in the
presence of weak base. Two intermediates [2] and [3] are thought
to be implicated in the mechanism. Suggest a possible structure for [2]
and mechanisms for the overall transformation, indicating any steps that might
involve the base.
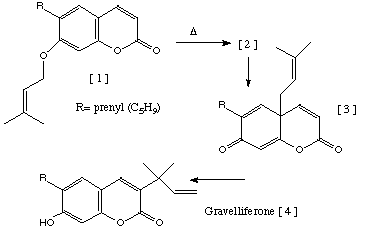
Answer
Qu 17. 1-Nitropropene when added slowly to a solution of phenyl isocyanate
and triethylamine (which together act as a dehydrating reagent) in the presence
of norbornadiene forms a mixture of two isomeric compounds A and
B. These both show three similar vinylic nmr peaks, but are
distinguished by different couplings in the non-vinylic region. A:
δ3.45 (J 8, 1.5 Hz), and 4.82 (J 8, 1.3 Hz) and B: δ3.87 (J
9.5, 4.2 Hz), and 5.29 (J 9.5, 4.3 Hz). Flash vacuum pyrolysis of
A+B yields cyclopentadiene and a new compound C, the
latter having the following 1H nmr spectrum: δ 5.61 (J 10.9,
0.9Hz), 5.90 (J 17.8, 0.9 Hz), 6.47 (J 1.7Hz), 6.8 (J 17.8, 10.9 Hz), 8.32 (J
1.7 Hz), which includes three vinylic and two aromatic resonances, and one
surprisingly low ortho aromatic coupling. Suggest structures for
A, B and C, mechanisms for their formation and an
interpretation of the different couplings manifested for A and B
(Hint: try using the MacroModel modelling program).
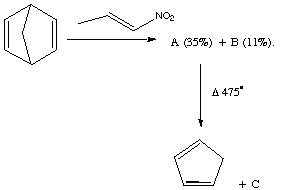
Answer
Qu 18. Random dot autostereograms are a way of representing three
dimensional images. Does this compound contain any chiral centres?
Answer
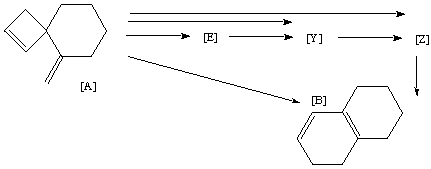
Qu. 19.
The transformation of [A] to [B] by heating has been extensively
studied kinetically. The evidence seems to suggest that direct conversion does
not occur. Synthesis of the triene [Z] suggests it is converted to [B] very
rapidly. Mechanisms have been proposed which indicate that whilst direct
conversion of [A] to [Z] is possible, it cannot be the exclusive pathway, since
an intermediate [E] is detected during reaction. [E] cannot convert thermally
to [Z] at the temperatures used, but would have to go via [Y]. It is also
possible that [Y] too is formed directly from [A], but this is thought less
likely on steric grounds.
Suggest possible structures for [E], [Y], [Z], mechanisms for all the possible
conversion routes shown with arrows below, and explain the cause of the steric
grounds referred to above.
Answer
Qu. 20. Reaction of two equivalents of the [1,3] dipolar species [S] with
pyridine gives a 43% yield of the adduct [P]. The 1H NMR of [P] is
δ 3.15 (dd, J 17, 6.8Hz), 4.00 (dd, J 17, 11.5), 6.20 (dd, J 11.5, 8.5),
6.42 (dd, J 11.5, 1.2), 6.45 (multiplet, J 11.5, 8.5, 6.8, 1.2), 6.8-8.4 (20H,
aromatics). Three intermediates [M], [N and [O] are thought to be involved.
Suggest possible structures for [N] and [P], together with mechanisms for their
formation, assignment of the NMR data for [P] and any stereochemical
conclusions derived from this data.
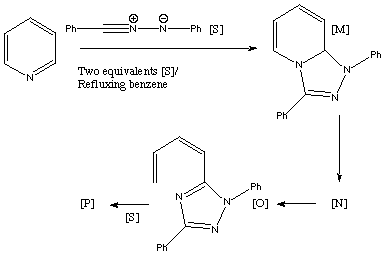
Answer
Qu 21.
When compound 1 is treated with base and heated, it rearranges to
give the products shown. The 13C isotope is distributed unequally
between two products. Explain this result in mechanistic terms, and clearly
indicate the type of reactions occurring and their nomenclature.
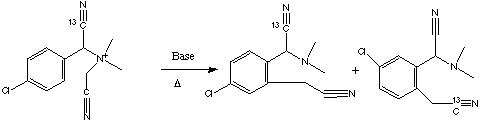
Answer
Qu. 22. Propose a mechanism for the following transformation.
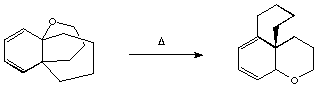
Answer
Qu. 23. Shown below are stages in the total syntheses of two interesting
molecules. Describe the type of reaction occuring at each stage, including
details of nomenclature and the number of electrons involved. Suggest
structures for the intermediates A, B and C, and propose
mechanisms for each stage.
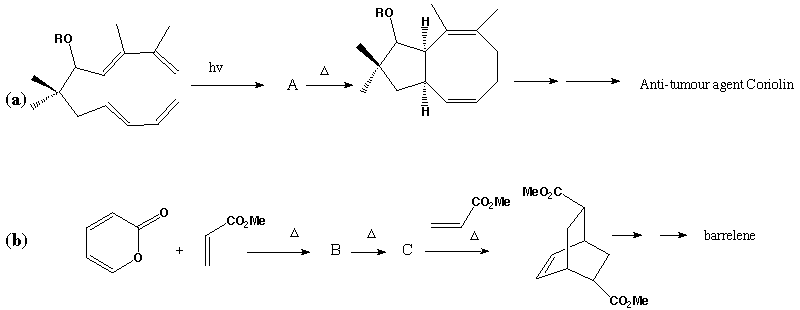
Answer
Qu. 24. PThe molecule below when heated gives rise to varying amounts of
the products D-H. Propose mechanisms to account for the formation of
each product. Some of these reactions will be pericyclic reactions, some (such
as the aromatising loss of hydrogen) will include steps that are not. Indicate
clearly which are the pericyclic steps.
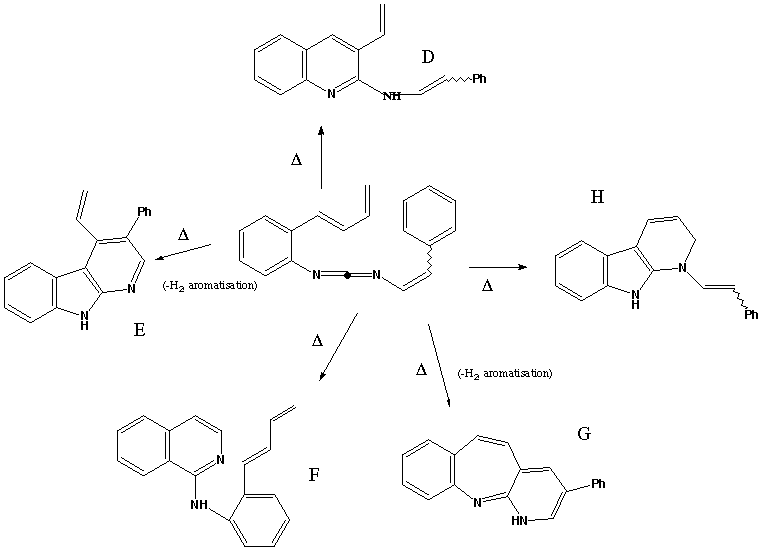
Answer