Nomenclature; pentose, hexose, etc. aldose, ketose; chemistry of the carbonyl group: via oxidation of aldoses to -onic acids and -aric acids; reduction; hydrazone and osazone formation; thioacetal formation; homologation (ascent) by the Kiliani method; degradation (descent) by the Weerman method; structural correlations by these methods
- Mutarotation of D-glucose - a and b pyranose and furanose forms in equilibrium. Pyranose form is usually favoured with most hexoses. Anomeric Centre
- Glycoside Formation (Glycosidation) under kinetic control both a and b methyl furanosides are formed. Under thermodynamic control the a pyranoside is the major isomer. The anomeric effect is explained in terms of lone pair-lone pair repulsion, dipole-dipole interactions and by M.O. theory. Glycosides are stable in neutral or basic solution but readily hydrolysed to the free sugar with dilute aqueous acid. Modern methods of glycoside synthesis: trichloroacetimidate activation.
- Reactions of Carbohydrates - Selective hydroxyl protection and neighbouring group participation
- Acetylation to give the a and b pyranose forms of the penta- acetate; preparation of a-glycosyl halides and their conversion to glycosides and formation of glycals; reactions of glycals e.g. with dilute aqueous acid to give 2-deoxy-sugars and with peracids to give epoxides
- Etherification
- MeI/NaH methyl ethers - difficult to remove
- Benzyl ethers - cleaved by H2/Pd or Na/NH3
- Silyl ethers - volatile for mass spectral studies, very readily cleaved by H3O+/or KF/H2O, variable reactivity depending on bulk
- Allyl ethers - acid stable, isomerisation with KOtBu or (Ph3P)3RhCl to give acid labile vinyl ethers
- Triphenylmethyl ethers, "trityl" ethers - selective for primary OH. Removed by dilute acid
- Acetal and ketal formation - ketones prefer to form 5 membered ring ketals-1,2 diol protection. Aldehydes prefer to form 6 membered ring acetals-1,3 diol protection.
- Sulfonate esters - preparation and selectivity for primary hydroxyl; intermolecular nucleophilic displacement reactions and their problems; deoxygenation via free radical chain reaction of xanthate esters with tri-n-buylstannane; intramolecular nucleophilic displacement reactions give ethers and epoxides (anhydro sugars); ring opening of epoxides and glycoside synthesis
- Keto sugars - best prepared by oxidation of alcohol with DMSO/Ac2O or DMSO/P4O10; nucleophilic attack from the least hindered side
- Unsaturated sugars - the Corey-Winter Olefin synthesis (thiocarbonates + trimethyl phosphite); reduction of ditosylates with sodium iodide and zinc
- Amino sugars - tosylate displacement with azide followed by LiAlH4 reduction; epoxide ring opening by ammonia; reduction of ketoximes with LiAlH4
- Use of Carbohydrates
- Fermentation
- Aminoglycosides are important in antibiotics e.g. erythromycin,
streptomycin.
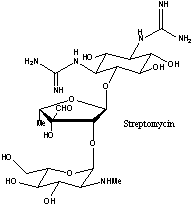
For a review of the preparation and reaction of aminoglycosides see: Topics in Antibiotic Chemistry Vol 1, p 1; Sammes, P.G., Ed.; John Wiley and Sons, 1977. - In non-carbohydrate natural product synthesis carbohydrates belong to the "Chiral Pool" i.e. they are a very cheap source of starting materials for organic synthesis which contain chiral functionality.