The H2 polymer
In this section you will calculate the wavefunction
and the band structure
for the hydrogen polymer with 2 atoms in the periodic cell, H2.
When the cell is doubled, a'=2 a,
the cell in the reciprocal space becomes half:
a*'= a*/2 .
The energy states that were in the range
between a*/4 and X=a*/2
in the band structure obtained at the end of Exercise 2
for the H polymer
are now folded back.
Exercise 1: Start DLVisualize,
run a CRYSTAL calculation for the H2 polymer:
H2_polymer.inp
Exercise 2: Run a CRYSTAL properties calculation
of the band structure for the H2 polymer,
as explained in Exercise 2
for the H polymer.
In the CRYSTAL Bands panel, type the coordinates of the
points
(note that the coordinates are expressed in unit of a*'):
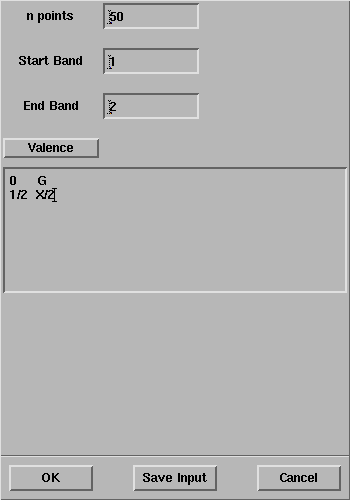
Questions:
- How many bands are visualised in the band structure?
- What are the differences between the current band structure
and the one
obtained for the H polymer?
- Draw the bonding and the anti-bonding combination for each band.
Index
Next

