Email Discussion:
Poster 30 (Raymond Jones)
Osman
Email Discussion:
Re: Poster No 30 (Ray Jones)
Simon Ellwood
Email Discussion:
30 (poster)
Raymond Jones
Email Discussion:
Re: Poster 30 (Raymond Jones)
Raymond Jones

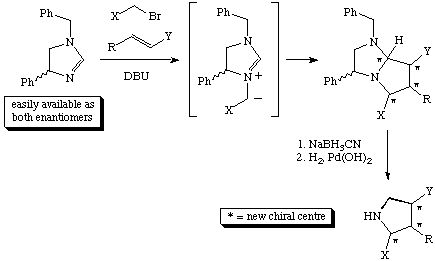
Scheme 1
During our development of this reaction, an unexpected result was observed.
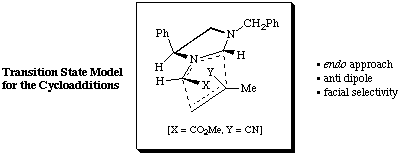
From the cycloaddition reaction between the homochiral azomethine ylide (S)-1 and methacrylonitrile two products (ratio 8:1) were obtained. It was expected1,2 from our model of the transition state for these cycloadditions, see below, that the two adducts would be the endo adduct 2 (major product) and exo adduct 3 (minor product), see Scheme 2.
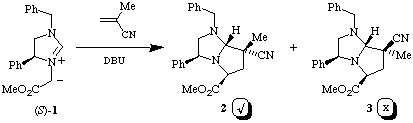
Scheme 2
1H N.m.r. experiments using n.O.e confirm the absolute stereochemistry of adduct 2 to be as expected; however the minor adduct was found not to be 3 but to have the absolute stereochemistry as shown in structure epi-3, i.e. epimeric at C-7a.
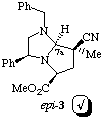
We assume that minor adduct 3 is formed initially (via exo approach to the anti-dipole configuration expected from our earlier work1) and undergoes subsequent epimerisation at C-7a to epi-3 during purification, for example by a route such as shown in Scheme 3.
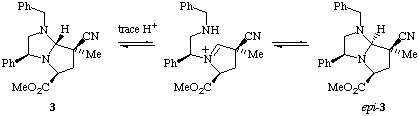
Scheme 3
Our rationale for this result assumes that epi-3 is more stable than primary exo adduct 3. Attempts to underpin this result using molecular modelling calculations have, however, proved unsuccessful to date.3 In all cases (we have several) where two adducts are obtained from the cycloaddition, and absolute stereochemistry has been determined, the isolated minor isomer has been found to be epimeric at C-7a to the expected exo adduct.
2. Jones, R. C. F.; Howard, K. J. J. Chem. Soc., Perkin Trans. 1, 1993, 2391-2393.
3. Molecular mechanics (MM2 or MM3, via Macromodel) and semi-empirical calculations (AM1 method, via Spartan) both have epi-3 as less stable than 3, in direct opposition to our experimental findings.
(corrected 07/07/95)