Introduction
In the search of new anticancer agents, we have developed a series of heterocyclic
compounds possessing:
1.- high affinity for DNA
2.- alkylating potential leading to IRREVERSIBLE BINDING to the
macromolecule.
To achieve our goal, we have synthesized different compounds in which a quinone-
methide or quinone-imine-methide precursor was carried by an intercalating heterocycle
(acridine or benzo[b][1,7]phenanthroline).
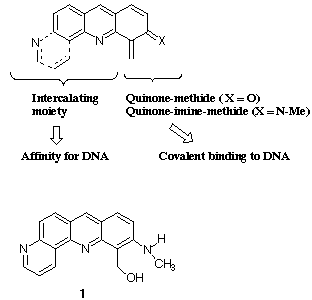 All compounds were tested on tumor cells both in vitro and in vivo (Collaboration with
Pierre Fabre Medicament Industry). One of the most cytotoxic compounds in vitro
appeared to be the alcohol derivative 1. This compound was also shown to be active but
toxic in vivo.
To explain the high activity of this molecule we have started a detailed study of its
chemical reactivity.
We found that compound 1 was reacting through two main pathways: a photochemical
degradation and an elimination mechanism leading to a quinone-imine-methide
intermediate. We have investigated these two pathways and we describe here our results.
All compounds were tested on tumor cells both in vitro and in vivo (Collaboration with
Pierre Fabre Medicament Industry). One of the most cytotoxic compounds in vitro
appeared to be the alcohol derivative 1. This compound was also shown to be active but
toxic in vivo.
To explain the high activity of this molecule we have started a detailed study of its
chemical reactivity.
We found that compound 1 was reacting through two main pathways: a photochemical
degradation and an elimination mechanism leading to a quinone-imine-methide
intermediate. We have investigated these two pathways and we describe here our results.


 All compounds were tested on tumor cells both in vitro and in vivo (Collaboration with
Pierre Fabre Medicament Industry). One of the most cytotoxic compounds in vitro
appeared to be the alcohol derivative 1. This compound was also shown to be active but
toxic in vivo.
To explain the high activity of this molecule we have started a detailed study of its
chemical reactivity.
We found that compound 1 was reacting through two main pathways: a photochemical
degradation and an elimination mechanism leading to a quinone-imine-methide
intermediate. We have investigated these two pathways and we describe here our results.
All compounds were tested on tumor cells both in vitro and in vivo (Collaboration with
Pierre Fabre Medicament Industry). One of the most cytotoxic compounds in vitro
appeared to be the alcohol derivative 1. This compound was also shown to be active but
toxic in vivo.
To explain the high activity of this molecule we have started a detailed study of its
chemical reactivity.
We found that compound 1 was reacting through two main pathways: a photochemical
degradation and an elimination mechanism leading to a quinone-imine-methide
intermediate. We have investigated these two pathways and we describe here our results.