CONCLUSION
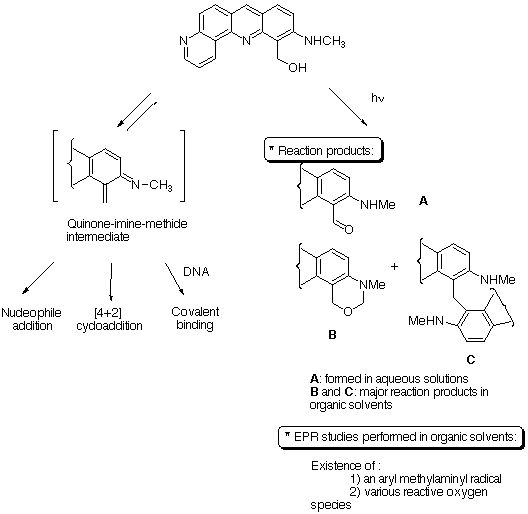
The methylamino-benzo[b][1,7]phenanthroline-methanol appeared to be a very complex molecule. It reacts photochemically to give reactive radical species, oxygen radicals and aryl methylaminyl radical. The formation of such species may be involved in the toxicity of the drug. In the absence of light, compound 1 is fairly stable, we have only observed minor degradation after one week of standing in organic solvents or in water. Il reacts with nucleophiles and with DNA. We propose that this reactivity is related to the formation of a highly reactive quinone-imine-methide intermediate. It should be noticed that in water the formation of the quinone-imine-methide is reversible, the nucleophilic addition of water gives the starting material. This reversibility can account for the high level of binding on DNA as the reaction with water is not a competitive reaction or a detoxication pathway as it is the case for most of the alkylating agents. The formation of a quinone-imine-methide carried by an intercalating heterocycle appeared to be very promissing in terms of selective DNA modifying agents. We are now working on other precursors and more water soluble agents.