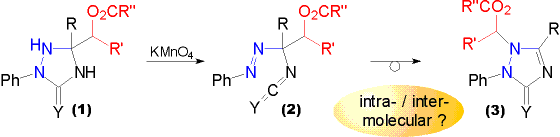
R = Me, Ph; Y = O, S

| In contrast to the hydroxyalkyl substituted triazolidin-3-ones and -3-thiones their acetyl and benzoyl derivatives (1) upon oxidation with potassium permanganate give isolable gem phenylazoisocyanates and -isothiocyanates (2), respectively. On standing they gradually rearrange to triazolin-3-ones and -3-thiones (3). | The former acyloxyalkyl substituent thus becomes part of an acylalaminal in the product structure (3). The question arose, whether the migration is intramolecular or intermolecular. To answer this question a special "CROSSOVER" EXPERIMENT was designed. |
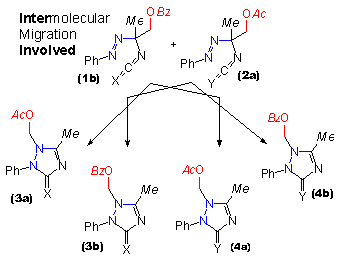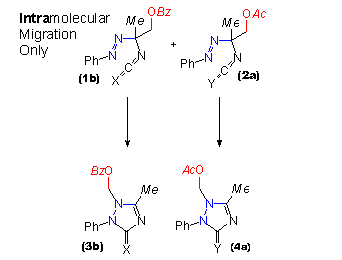
| The design of this crossover experiment makes use of the similarity between triazolidinones and their sulfur analogues. Yet all different intermediates in the course of the oxidation reaction can be distinguished by nmr after they had been prepared separately before the experiment. In two distinct experiments pairs of the acylated compounds were taken (one triazolidin-3-one and one triazolidine-3-thione of which one was acetylated and one was benzoylated) and they were oxidized together. Thus a mixture of an azoisocyanate and an azoisothiocyanate (1b, 2a) was obtained. The slow rearrangement was monitored by nmr spectroscopy. |
In this animated scheme both product distributions of triazolin(thi)ones expected for the intramolecular migration (3a, 3b, 4a, 4b) and for the intermolecular migration (3b and 4a only) are shown as alternatives. In the case of intermolecular migration in addition to the products of intramolecular migration, compounds with interchanged acyl residues (3a and 4b) are to be expected. In the experiments performed within the experimental limits only signals according to the products of strict intramolecular migration were found. |
| TOPICS OF: | F) OXIDATION HYDROXALKYLTRIAZOLIDINONES: |
| F1) General Reactivity F1.1) Crossover Experiment NEXT chapter HOME INDEX |