Mechanistic proposals
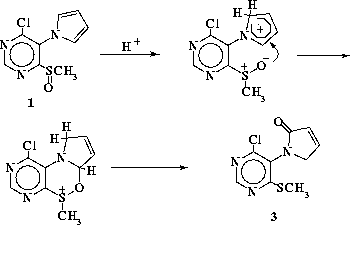
We propose two possible reaction pathways to account for pyrrolinone formation
during SES cyclization (Schemes A and B). Scheme A is based on chemistry much more
common for furans than pyrroles. [4] Scheme B relies
on the well known oxidizing power of sulfoxides [5],
although we have found no literature precedence for the oxidation of pyrrole by sulfoxides. Hydrolysis of the initially formed trifluororacetate ester in this pathway to the observed
pyrrolinones under the prolonged alkaline work-up conditions would not be surprising.
Scheme A
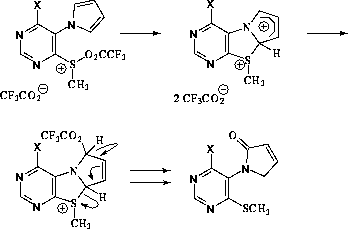
Scheme B

 Back to abstract
Back to abstract




 Back to abstract
Back to abstract